// FRESENIUS KABI Case Study
SERIALIZATION DRIVING INNOVATION & CHANGE WITH RFID
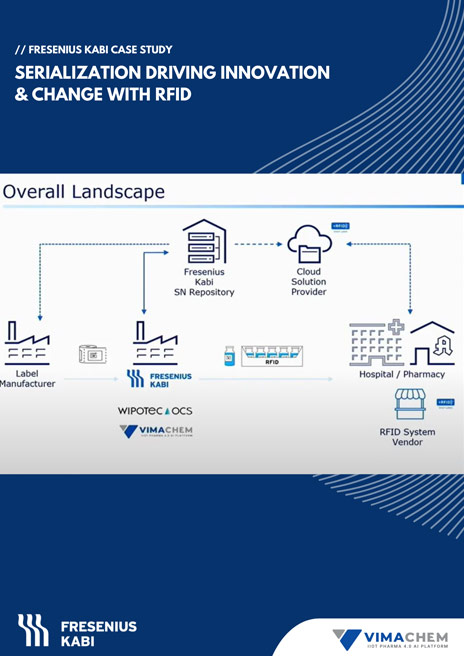
Fresenius Kabi to optimally support the US healthcare system, decided to implement RFID (Radio Frequency Identification) inventory tracking systems for pharmaceutical products sold in US hospitals. Without RFID, the process of identifying what products are in the hospital and how many, is very manual, repetitive and time consuming. With RFID tagging, the process of identifying and counting Fresenius Kabi products in US hospitals, is automated and compliant allowing Fresenius Kabi to optimally support its healthcare customers.
Challenge: Manual tagging is time-consuming, labor-intensive, and possible errors.
Benefit: Accurate/ ready-to-read, compliant and interoperable.
Fresenius Kabi Products with RFID tags
- Ready-to-read, interoperable, RFID-embedded
- Label/tag specific to the product and batch
- Standardized format to enable interoperability (GS1)
- Direct-read or cloud-compatible
- Rain RFID
- Axia-Lab tested
Key Learnings
- RFID and data matrix GS1 codes, complement each other and are used in different ways in various clinical workflows.
- RFID provides additional benefits of minimal handling and wireless identification.
- Required patient documentation at the point of dispense can be automated (e.g., in case of controlled substances handling).
- Manufacturer’s RFID solutions should be based on open standards to enable interoperability (e.g., GS1, RAIN).
- During the design and implementation phase new (and surprising) obstacles may be faced.
- Range of RFID enabled products is growing.