Testimonials
// View our clients testimonials
What our Clients say
We are fortunate to have formed excellent partnerships with our clients.

Michael Hadjion
Engineering Manager | Aspen Pharma Australia
"Aspen Pharma (Australia) are utilising Vimachem SSM as the site level serialisation system, which is paired with Wipotec OCS line level units. Vimachem SSM has a very clean, intuitive interface which allows new business users to be trained easily and to quickly gain proficiency in site serialisation operations. Where support is needed, the Vimachem team are quick to respond. Overall, Aspen have been highly satisfied with the Vimachem SSM application and highly recommend its use for companies that are going down the path of Wipotec-OCS as their serialization machines."

Dr. Reiner Pöstges
Head of Production / QP | MEDICE Arzneimittel Pütter GmbH & Co. KG
"At Medice we selected the Vimachem Level 3 Serialization Site Manager (SSM) to manage our plant level serialization operations, to manage our Wipotec-OCS lines (full aggregation) and to integrate to Tracelink L4-5. From the day of the kick-off of the Vimachem L3 project and for the 2 months until its complete installation and validation, we were amazed by the quality of the Vimachem software as well as with the expertise and professionalism of the Vimachem team. Vimachem supported us throughout the project with technical expertise and consulting for both the Vimachem L3 as well as for Tracelink L4-5 which was of great help to us. They also executed with us the full validation of the Vimachem L3 which combined with the high quality documentation of Vimachem, allowed us to timely install and validate the Vimachem L3 SSM in the Medice systems. We had a great experience working with Vimachem , we highly recommend the Vimachem L3 SSM and the Vimachem team."

Meghana Kalluri
Aseptic Manufacturing Engineer | Athenex Inc.
"Vimachem platform is extremely user friendly, required minimal training for Athenex team. Vimachem project implementation team was extremely supportive, especially towards the rush hour and prior to launch. Their qualification documentation and approach were very customer specific and less generic, which was helpful to narrow down our internal testing. The project team was on top of the schedule, always accessible through emails and supported fast paced execution to meet internal production needs."

Luis Montero
Engineering and Information Systems Director | IBERFAR, Indústria Farmacêutica S.A.
"Order generation and the connectivity to the lines (easy to add, change order from and to machine) was one of my favorite features. The Project team was quick to resolve and meet requirements throughout the project implementation, which had a duration of 2 months as promised before the project start."

Per Stenlöf
Project Manager | AGB Pharma AB
"The system is simple in use but can manage several complex settings. A highly valuable tool as it is very simple to easy and when compared to other similar systems. It only required one training session before we all were up and running.
What we really liked was the integration with TraceLink and how easy it was to reuse the master data. This minimizes the risk of using incorrect master data together with the master data verification. We also enjoyed that you could verify and preconfigure production lines and templates to minimize risk´s related to master data entries using the test environment. The option to automate commissioning and product release features is useful and effective to less user input while staying compliant.
Support and access to the system is easy, maintaining a high level of security and data integrity. Our project was installed on time and the project team was very competent with a very high level of support, competence which is key to understand the GxP requirements from any Pharmaceutical company.We also purchased the PQ validation from Vimachem to benefit from their experience. The validation documents kept a very high level of quality. All the work was made remotely due to the pandemic but the transition and execution were excellent.
During the installation project we also used the line simulator – an environment that can easily connect to the SSM QA and function as a real packaging machine. Very useful in integration tests against the real packaging lines for testing.Regular project meetings, either weekly or biweekly depending on our needs maintained good project team spirit and progress through project tasks on time. The Vimachem team approach was team oriented. Everyone attended the project meetings as much as they could and were active and supporting project progress. Even if we experienced a change in project lead during the execution of the project, the handover was barely noticeable.
I strongly recommend Vimachem."
What we really liked was the integration with TraceLink and how easy it was to reuse the master data. This minimizes the risk of using incorrect master data together with the master data verification. We also enjoyed that you could verify and preconfigure production lines and templates to minimize risk´s related to master data entries using the test environment. The option to automate commissioning and product release features is useful and effective to less user input while staying compliant.
Support and access to the system is easy, maintaining a high level of security and data integrity. Our project was installed on time and the project team was very competent with a very high level of support, competence which is key to understand the GxP requirements from any Pharmaceutical company.We also purchased the PQ validation from Vimachem to benefit from their experience. The validation documents kept a very high level of quality. All the work was made remotely due to the pandemic but the transition and execution were excellent.
During the installation project we also used the line simulator – an environment that can easily connect to the SSM QA and function as a real packaging machine. Very useful in integration tests against the real packaging lines for testing.Regular project meetings, either weekly or biweekly depending on our needs maintained good project team spirit and progress through project tasks on time. The Vimachem team approach was team oriented. Everyone attended the project meetings as much as they could and were active and supporting project progress. Even if we experienced a change in project lead during the execution of the project, the handover was barely noticeable.
I strongly recommend Vimachem."

Carlos Gancho
DSI - SA - System Administrator | Edol
Thank you very much for your invaluable help. Once again, you have been incredibly supportive-far better than our L4 provider's support. We truly need more business partners like you. Thank you once again for your amazing support.

David Tixier
Head of IT | Astrea
It's light for Astrea team to implement OEE, deployment is easy.

George Vlassopoulos
Head of Engineering | Elpen
Our journey with Vimachem has been truly rewarding. From connecting machines to installing the Vimachem MES platform, we rapidly integrated 6 packaging lines without any production disruptions.
• Elpen Case Study
• Elpen Case Study

Eleftheria Pekopoulou
Plant Director | Elpen
After thorough market research, we opted for Vimachem... Their specialization in developing pharma focused digital solutions fulfilled our specific needs and objectives. We were drawn to the user-friendly interface and streamlined maintenance of the Vimachem solution which made it an ideal fit for our operations.
• Elpen Case Study
• Elpen Case Study

David I
Head of Packaging | CDMO
Reliable OEE Partner
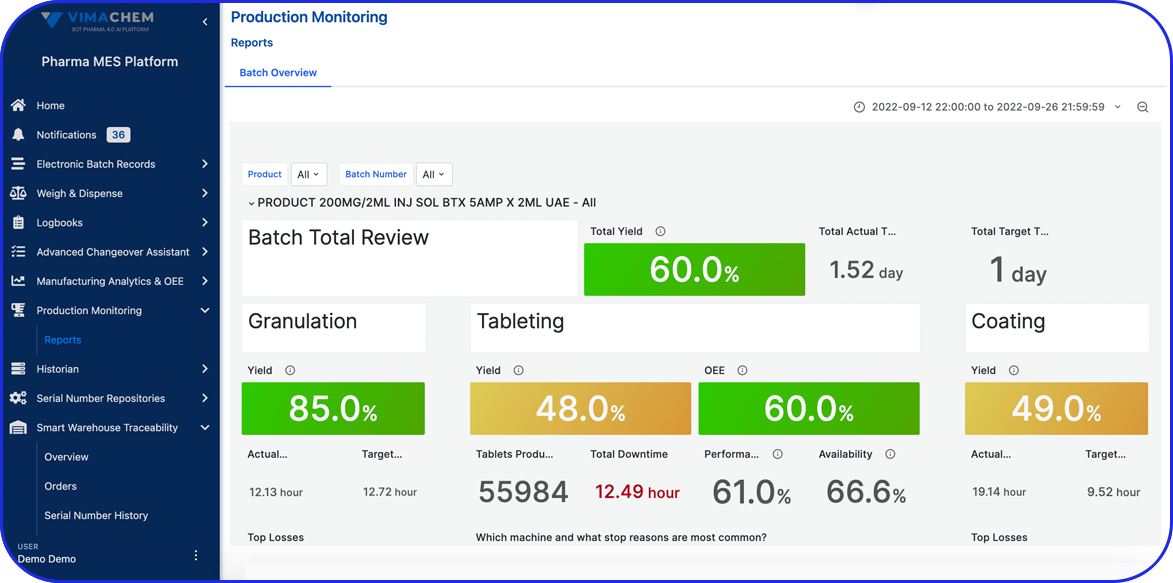
The platform offers strong IoT integration with machines, robust reporting, and a wide range of configuration options. The Vimachem Pharma MES Platform is helping us eliminate manual data entry and gain real-time visibility into production performance.
• Read more on G2
The platform offers strong IoT integration with machines, robust reporting, and a wide range of configuration options. The Vimachem Pharma MES Platform is helping us eliminate manual data entry and gain real-time visibility into production performance.
• Read more on G2
CSV & Serialization Manager
Pharmaceutical Manufacturing
Your implementation and support services are excellent, and we’re very happy with the experience so far.
As someone new to Vimachem and MES in general, I've found your team's support incredibly valuable, every interaction has significantly helped me, and I truly appreciate the pharma-specific expertise your team brings to every conversation.
As someone new to Vimachem and MES in general, I've found your team's support incredibly valuable, every interaction has significantly helped me, and I truly appreciate the pharma-specific expertise your team brings to every conversation.

David Tixier
Head of IT | Astrea
No change control, no downtime. We connected remotely to our existing data and had the first version running in weeks.
• Astrea Case Study
• Astrea Case Study

David Tixier
Head of IT | Astrea
Now even when our managers are traveling, they know instantly how the lines are performing.
• Astrea Case Study
• Astrea Case Study

Aurore Berthelemy
Operational Excellence Lead | Astrea
It works on its own now. I don't need to spend time on it - it's become part of how we work. And the system doesn't just measure - it helps us improve every week.
• Astrea Case Study
• Astrea Case Study

Anabela Claro
IBERFAR Operator from the Weighing Operations
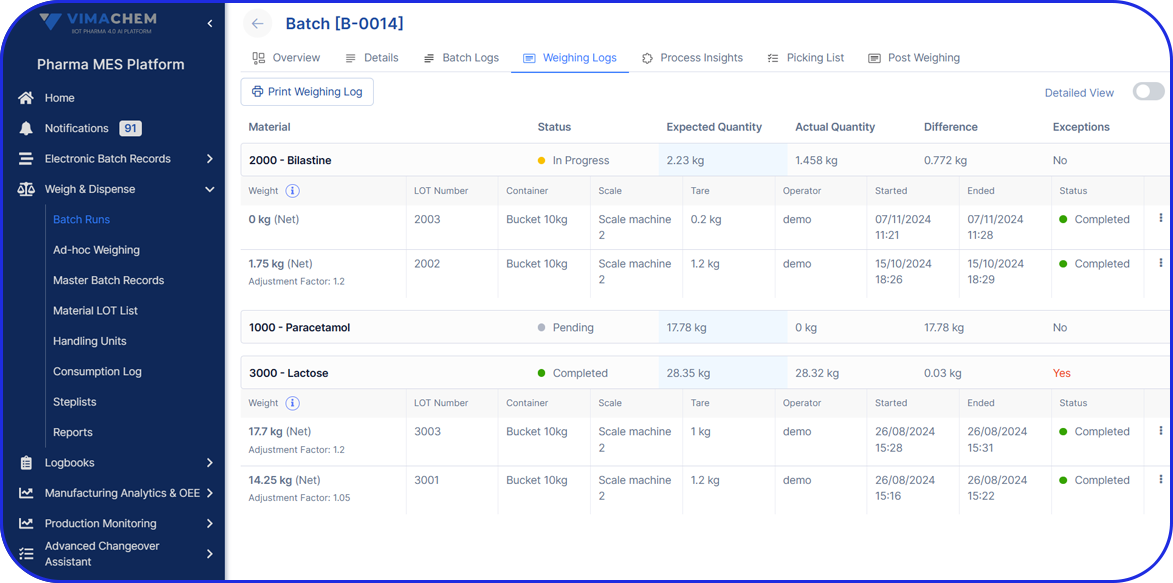
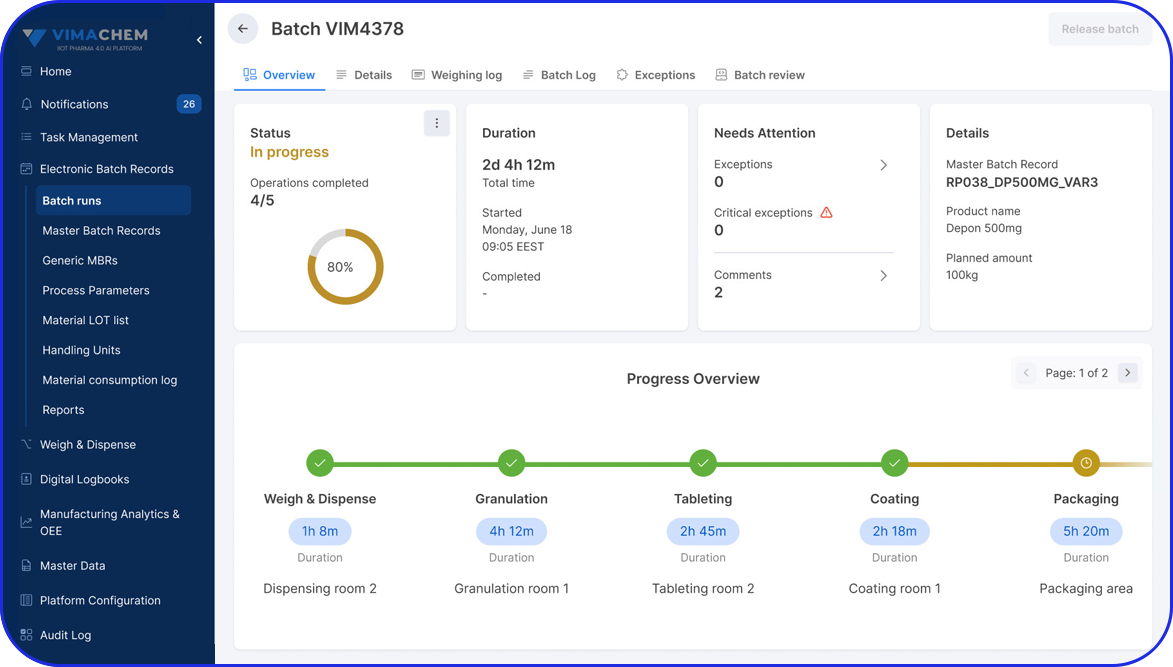
With the system, I no longer waste time on manual paperwork. It’s intuitive, easy to use, helps me keep track of the progress and guides me step by step through the process.
// partner testimonias
Discover how Vimachem’s long-standing collaborations with an array or partners supports pharma manufacturers in accelerating their journey toward fully integrated, data-driven manufacturing.
// Customer Testimonial
Learn how digitalizing manufacturing processes with Vimachem improves visibility, performance, and operational control across the shop floor. By connecting production data and enabling real-time insights, Vimachem helps teams optimize efficiency, reduce downtime, and support continuous improvement.