Digital Work Instructions & Checklists Software for Pharma Industry
Designed solely for Life Sciences, Digital Work Instructions and Checklists kickstart your digital transformation process and enable the right-first-time execution of daily operations.

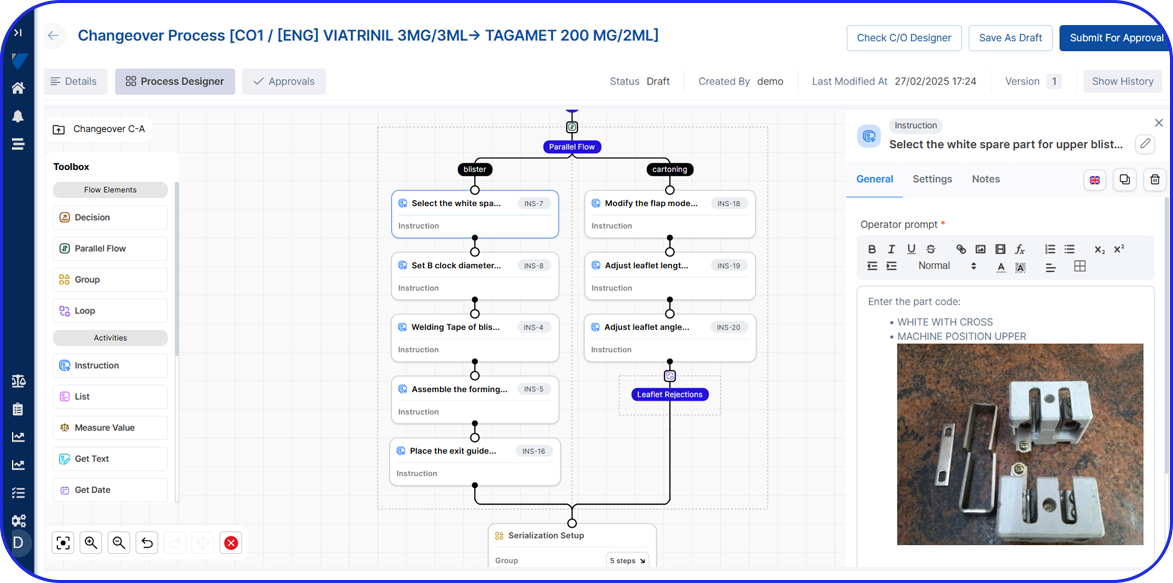
Standardize Pharma Execution With Digital Work Instructions
Digital SOPs
Automate and streamline processes, reducing the time it takes to complete tasks while ensuring data integrity and minimizing the risk of non-compliance.
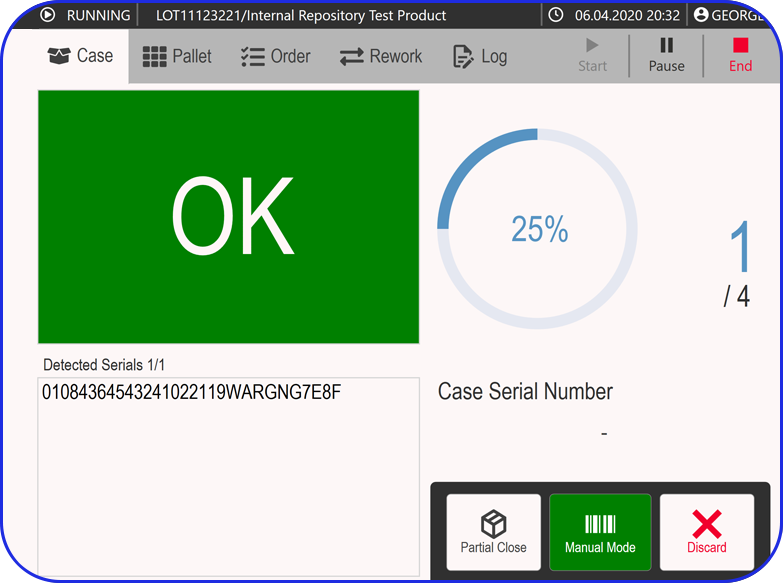
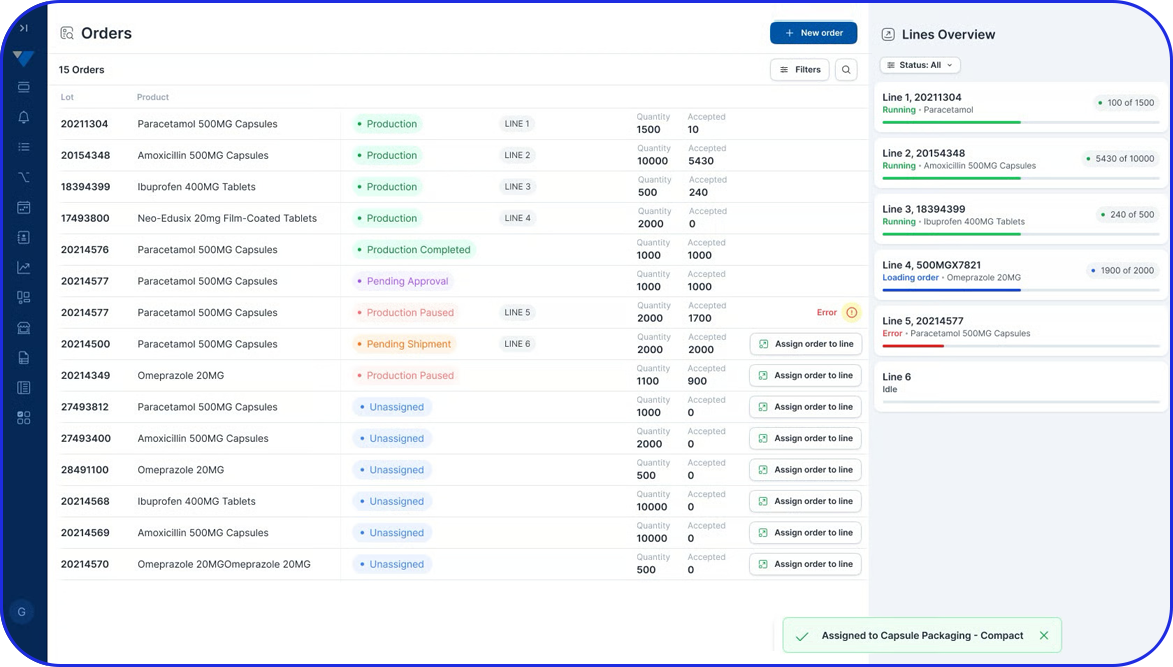
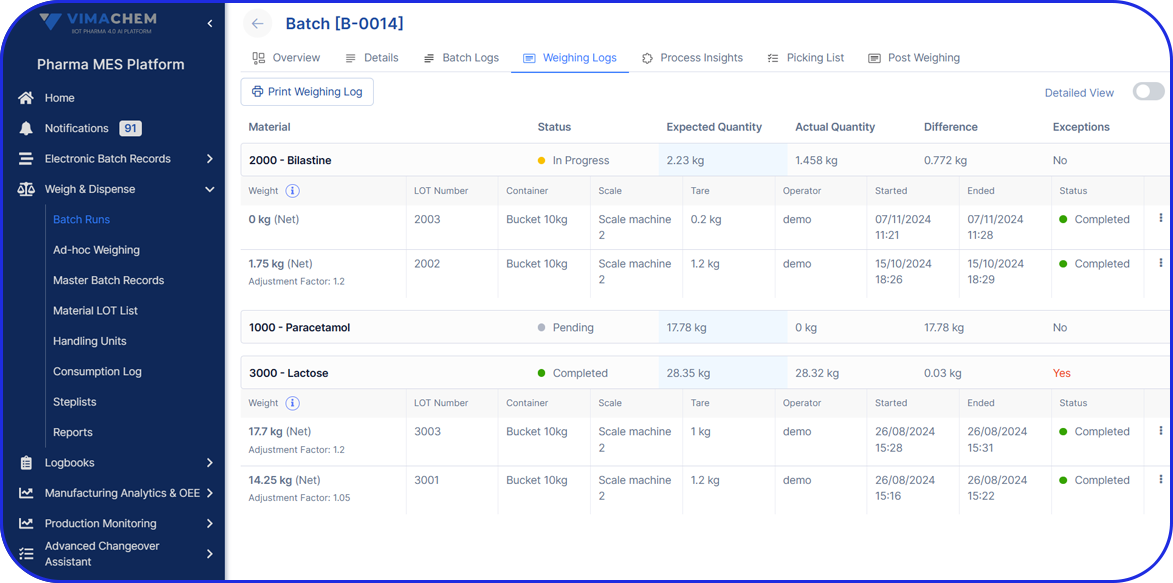
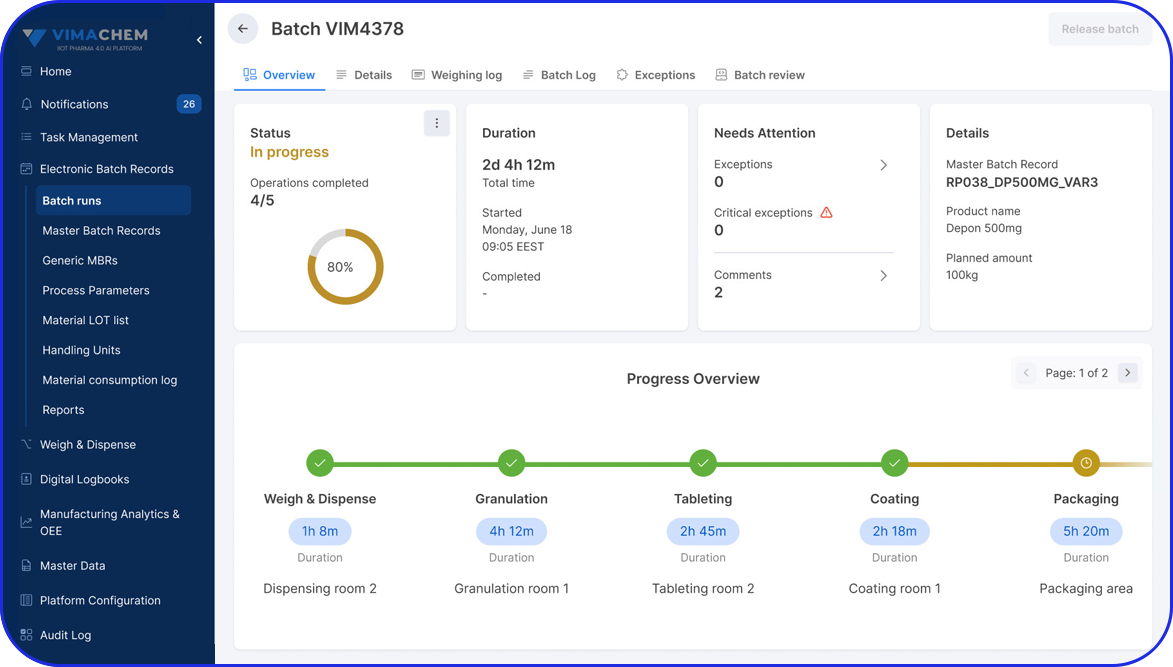
Electronic Batch Record
Use your checklists to enrich electronic batch records by guiding users through workflows and eliminating documentation errors.
Line Clearance
Streamline compliance with regulatory requirements and ensure efficient processes and finished product quality.
Training Management
Provide training consistently and to a high standard, reducing the risk of non-conformities and variability in performance.
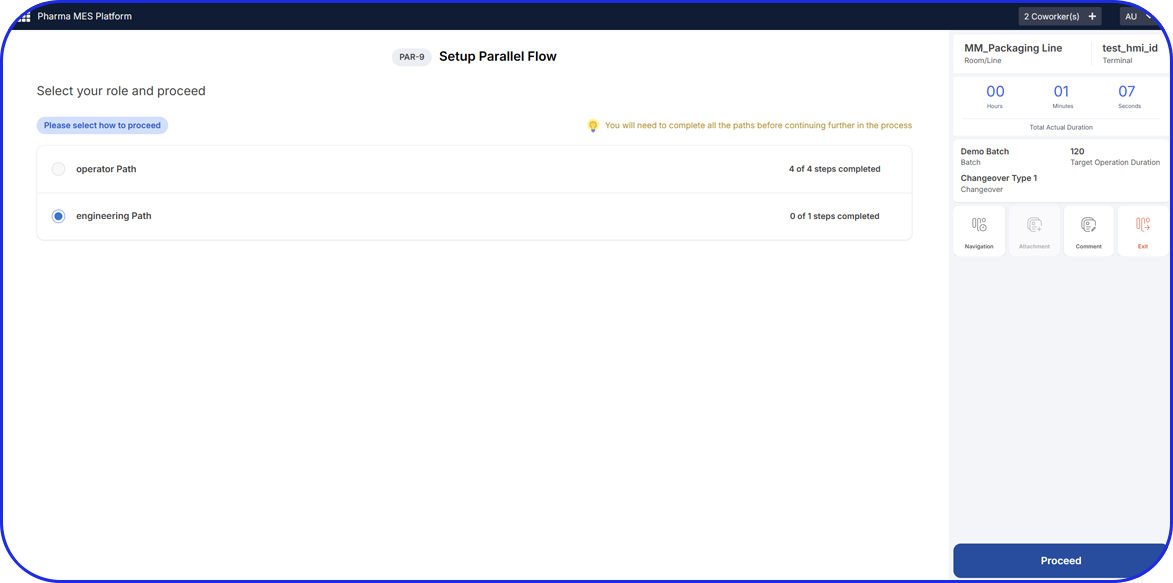
Process Management
Maximize efficiency, minimize risk, and ensure compliance with total flexibility of workflows specifically designed for pharma manufacturers.
See Digital Work Instructions & Checklists Software in Action
Digital Work Instructions & Checklists Software Built for Pharma
Built exclusively for pharma and biopharma, Digital Work Instructions (DWI) and Checklists easily adapt to your pharma needs, significantly reducing implementation/validation and training effort, allowing rapid go-live and easy maintenance.
The benefits, combined with the support of our experienced consulting team, help guide you during this digital journey with guaranteed success.

Digital Work Instruction Software Capabilities for Pharma Manufacturing
- Designed to meet the regulatory compliance requirements, including FDA, GMP, and FDA Title 21 CFR Part 11, to ensure data and processes are secure, validated, and auditable
- Coming with an out-of-the-box validation package (including IQ/OQ), which saves at least two months of effort during the implementation period
- Flexible enough to adapt to changing regulatory requirements, new products, and process changes without requiring significant customization
- Scalable and able to handle the increasing demands of a growing pharmaceutical manufacturing operation, including multiple sites and global operations
- Integrating with IoT devices such as smart sensors and wearables to provide real-time feedback on critical parameters and improve the data quality
- User-friendly, including capabilities to attach photographic evidence at any step of the process and including media instructions whenever necessary
- Being device-agnostic allows for seamless and flexible usage across multiple devices, leading to increased accessibility, user adoption, and productivity
Benefits of Digital Work Instructions Software for Manufacturing

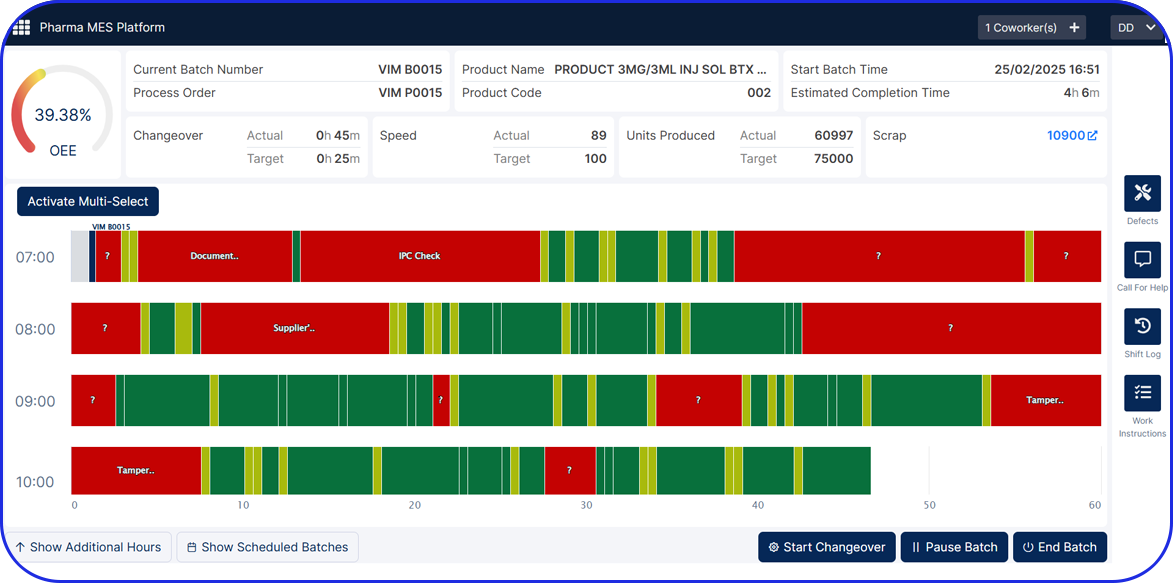
Gain real-time visibility
Track process step time for every step and get accurate and timely insights into all your operations, enabling quick data-driven decision-making.

Increase traceability
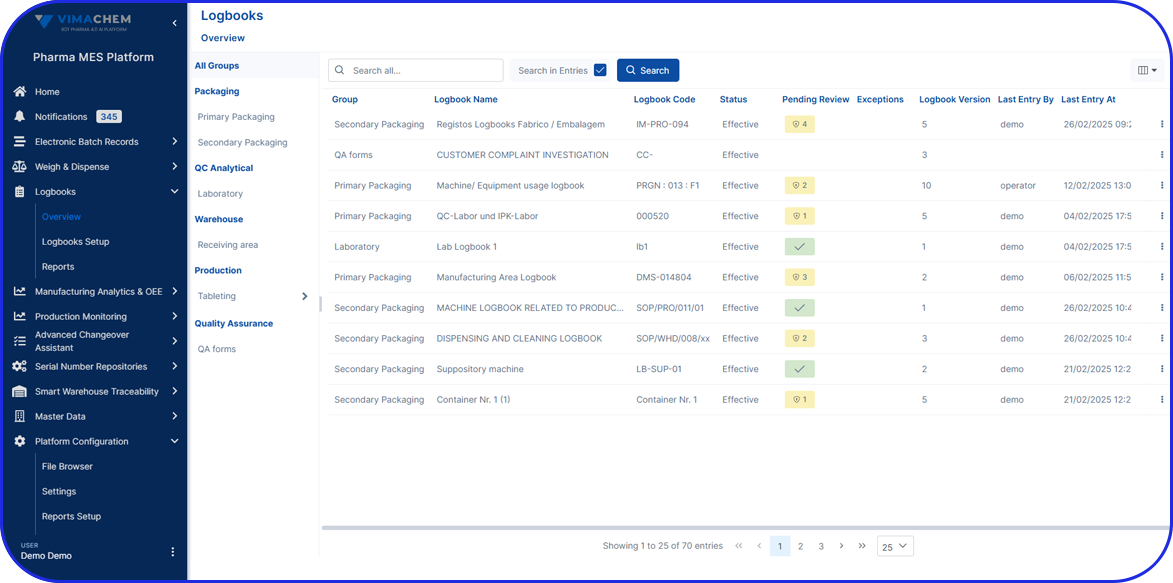
Quickly trace actions and events in the production records. Use the evolved audit trail to easily retrieve information by filtering, ordering, and organizing it in different ways.

Make shopfloor error-proof
Easily configure pre-requisite and format enforcement to minimize human error. Make right-first-time execution of the standard with non-conformance alerts and programmable guardrails that prevent operators from incorrectly completing a step when values diverge from defined ranges.

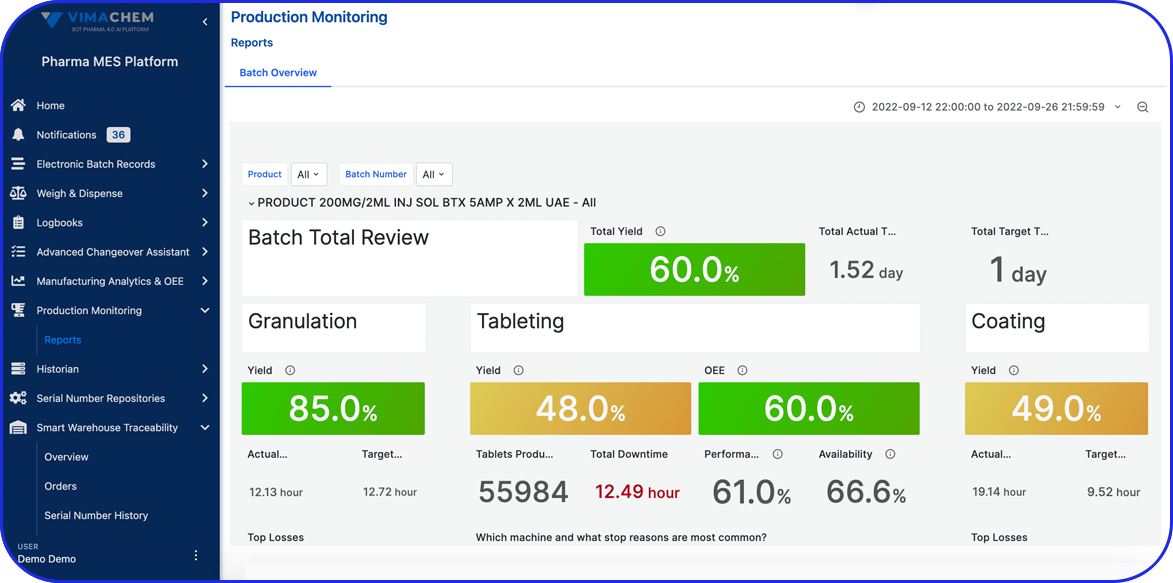
Use your advanced reporting and dashboards
Allow users to view the information that is most important to them in a comprehensive visual format to allow insights for improvements.

Eliminate bottlenecks
Monitor process completions in real-time to detect and resolve bottlenecks faster.
Design digital procedures in minutes
Rapidly digitize existing work instructions with an automated AI-based designer that converts existing PDF to digital work instructions in minutes.

Enrich your instructions and procedures with the help of media
Enhance instruction comprehension and increase engagement with the use of videos and photos to turn complex flows into seamlessly understandable ones.
Enhance collaboration
Execute together or assign individual steps to different users with a tool that promotes seamless communication and efficient workflows resulting in the streamlining of operations like never before.
GxP Features Built for Pharma Digital Work Instructions
User-Level Permissions & Security
Manage roles and permissions to keep data safe. User administration in a Windows Active Directory simplifies the definition of password criteria, reset and change periods, and reduces the amount of IT support.
Data Integrity
System fitness for purpose with verified system inputs and controlled access.
Electronic Records & Signatures
Fully compliant with FDA Title 21 CFR Part 11 and EU GMP Annex 11 (Electronic Records and Signatures).
Digital Records
A new way to manage the data that matters the most for compliance, including device records and equipment logs.
Electronic Signatures
Enable efficient review and approval through configurable workflows.
Data Exports
All data can be exported both manually and automatically in Microsoft® Excel® (XLS) format as well as Extended Markup Language (XML) format.
FAQs on Digital Work Instructions
What are Digital Work Instructions?
Digital Work Instructions (DWI) are step-by-step, guided procedures delivered through software. They replace paper or static SOPs with interactive workflows that enforce the right sequence, required fields, and quality checks, while capturing timestamps, evidence, and a full audit trail.
What is digital work instruction software?
Digital work instruction software is the platform used to design, deploy, and manage DWIs across teams and sites. It typically includes workflow design, permissions, audit trails, reporting, and integrations so execution is standardized, traceable, and scalable.
What’s the difference between digital work instructions and digital checklists?
Digital checklists confirm that tasks were completed. Digital Work Instructions guide how tasks should be completed, step by step. In practice, they work best together: the instruction drives execution, and the checklist captures required checks, approvals, and proof of work.
How do digital work instructions support GxP and GMP compliance?
A GxP-ready digital work instructions solution helps standardize execution, enforce required steps, and record every action in a secure, auditable way. It supports compliance by controlling access, strengthening data integrity, and maintaining complete records for inspections and reviews.
Can digital work instructions enrich Electronic Batch Records?
Yes. Digital checklists and DWIs can guide users through batch steps, capture timestamps and required values, and reduce documentation errors. The result is cleaner execution data that strengthens the batch record and speeds up review.
Can digital work instructions scale across multiple sites and global operations?
Yes. A scalable DWI solution supports standardized procedures across sites while allowing controlled variation where needed. This makes it easier to roll out new products, adjust workflows, and maintain consistent execution as operations grow.
Ready to Augment your Shop Floor Operations?
Ready to Augment your Shop Floor Operations?
Get started with real-time manufacturing analytics today.