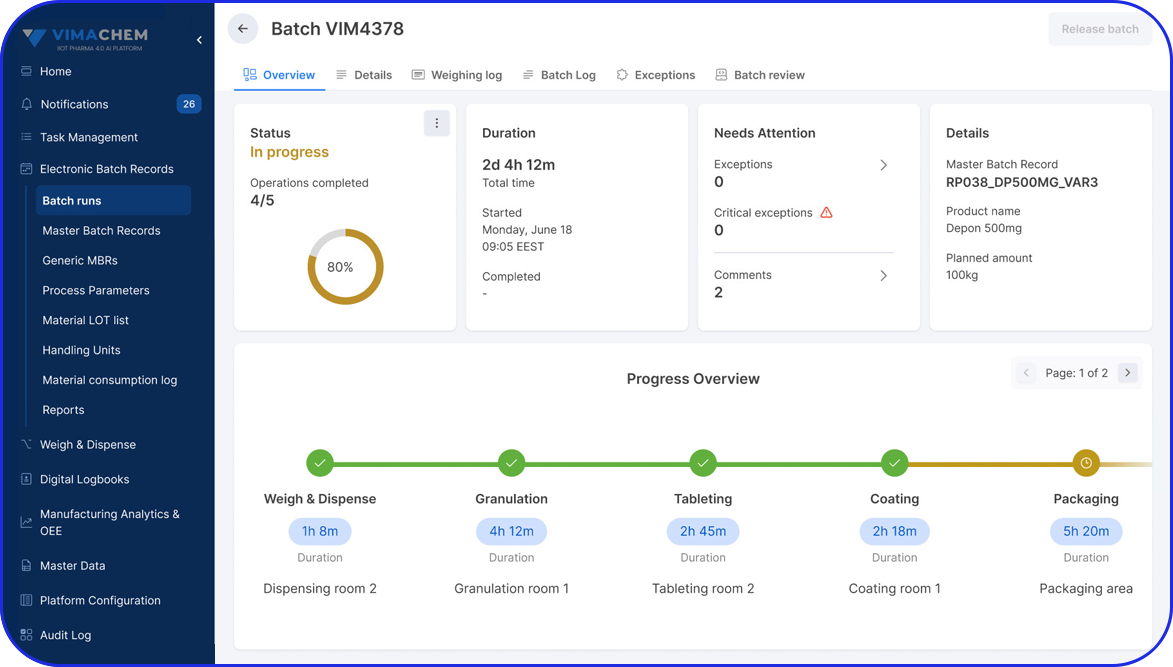
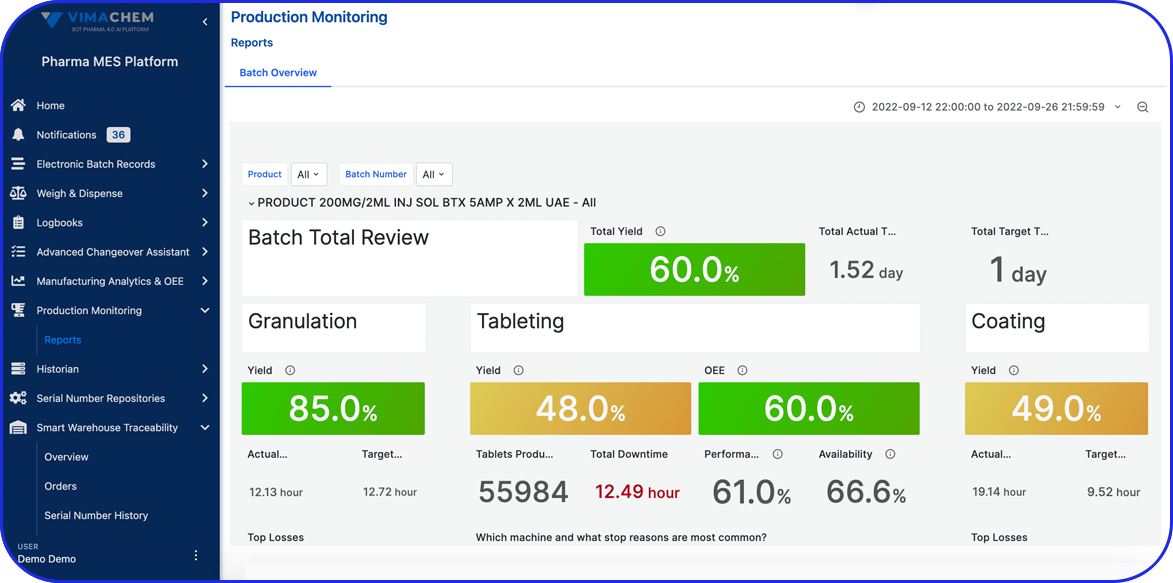
Manual Serialization & Aggregation Module
Add a compliant manual serialization and aggregation station to your Vimachem Level 3 SSM for low volume serialization, offline workflows, and post-batch rework. With a scanner, printer, and PC-based operator workstation, manufacturers can serialize and/or aggregate packaging levels and maintain full traceability across existing Level 2 and Level 4 connections.
See Vimachem’s MAM in Action
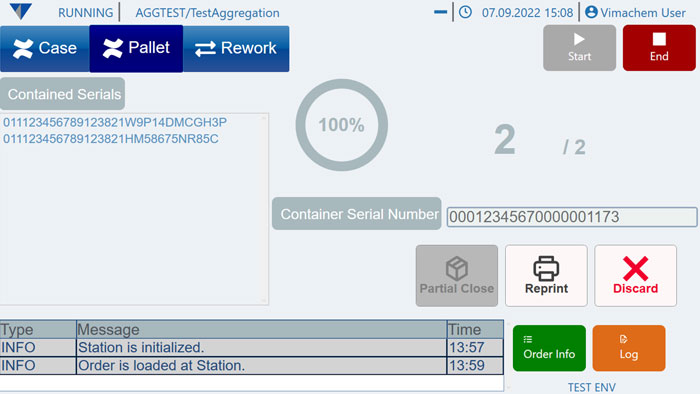
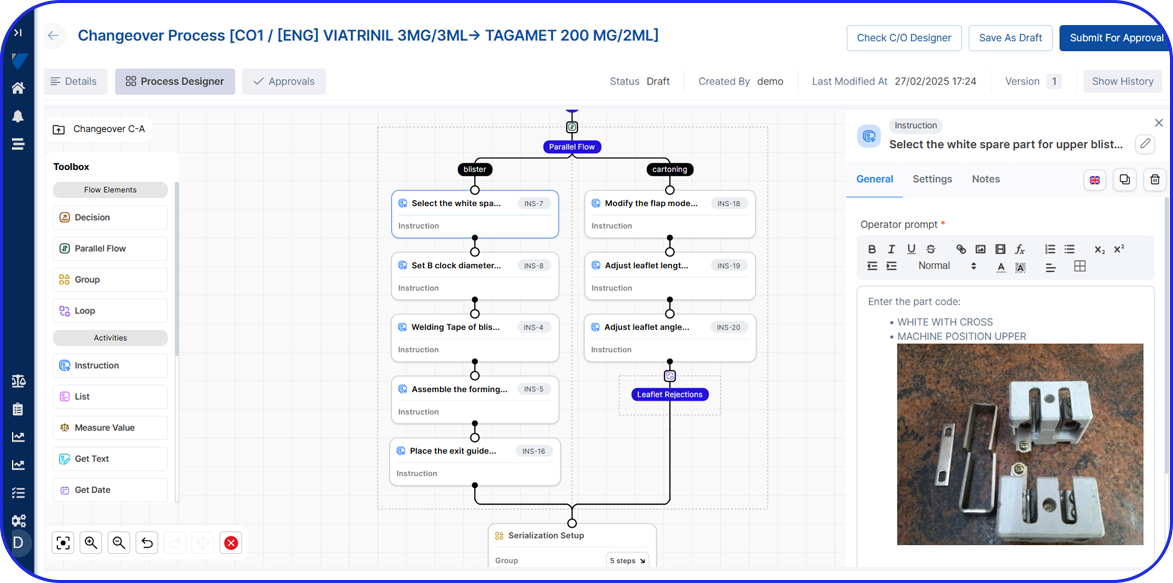
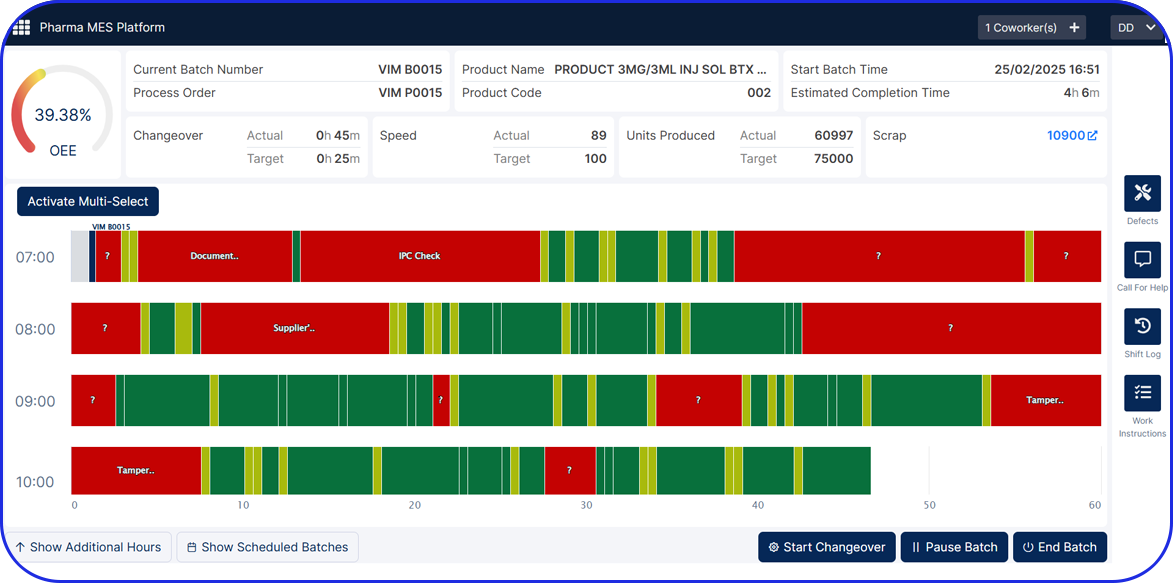
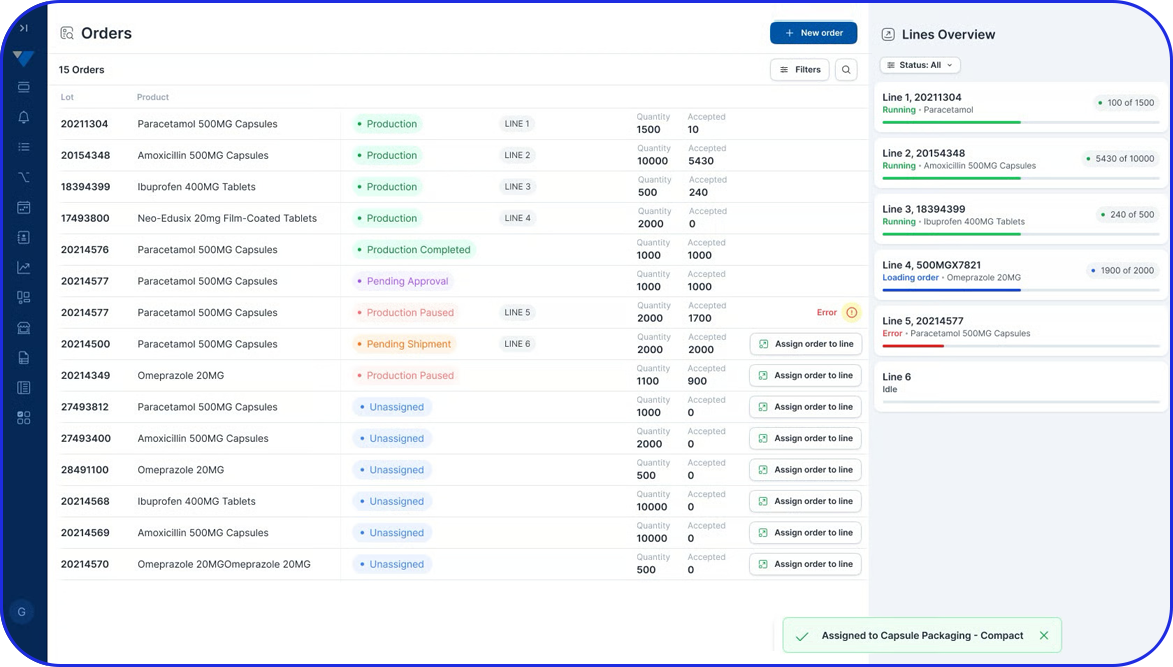
Manual Aggregation Module UI
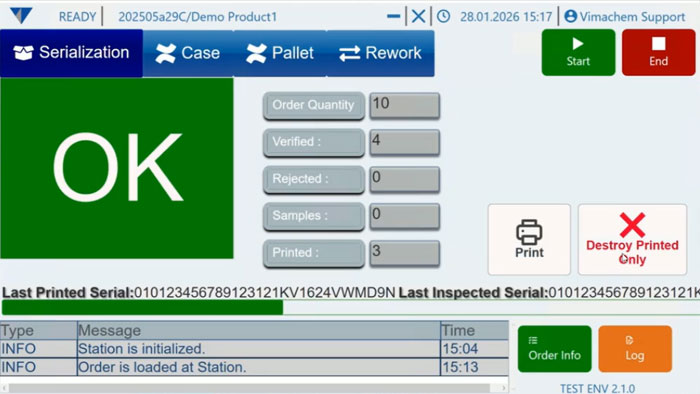
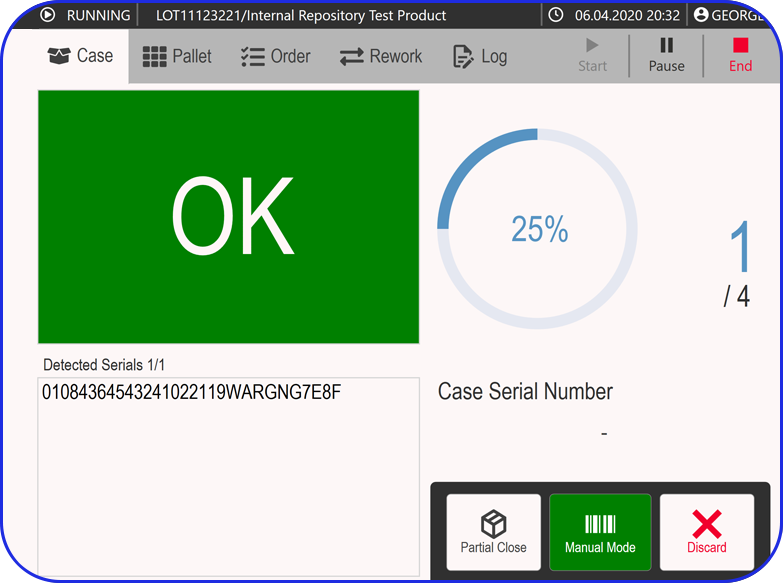
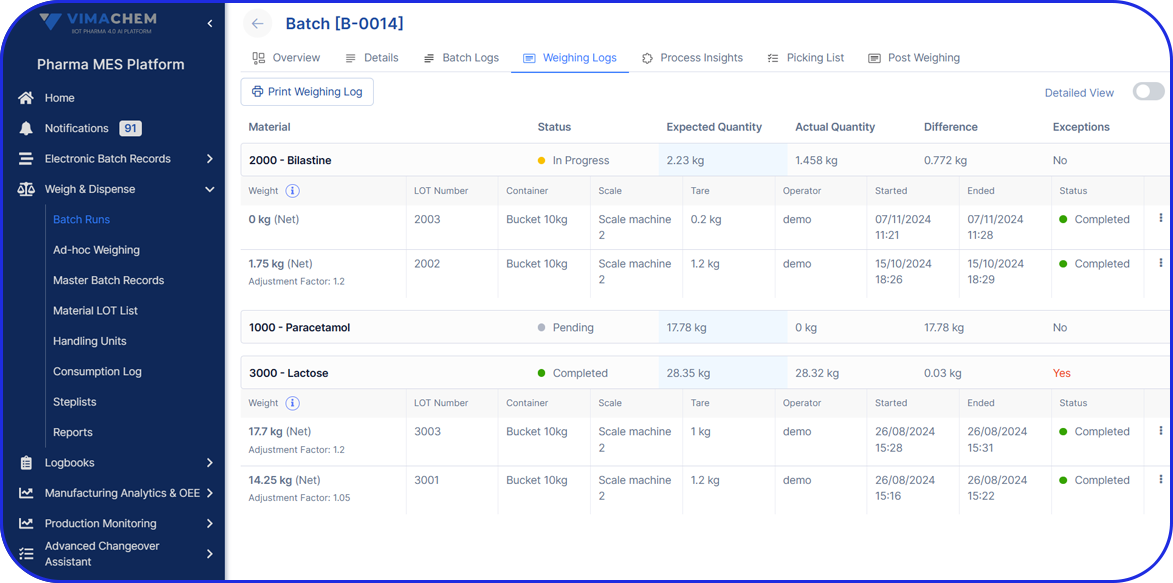
Manual Serialization Module UI
What is the Manual Serialization and Aggregation Module (MSAM)?
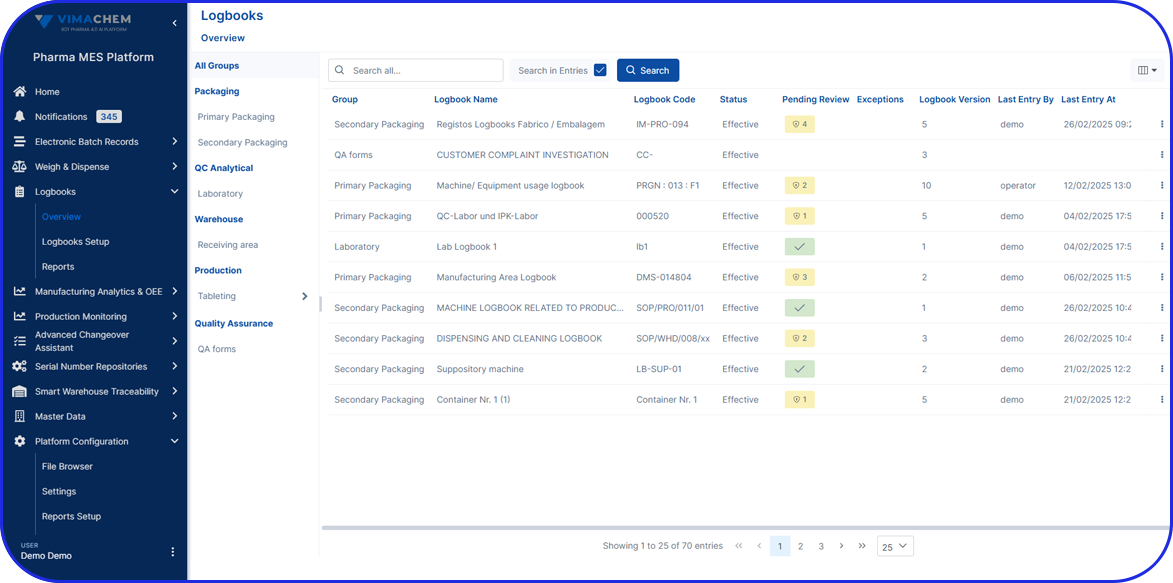
The Vimachem Manual Serialization and Aggregation Module (MSAM) is a modular, pharma-specific workstation solution for manual serialization, manual aggregation, and manual rework. Designed as an add-on to Vimachem’s Level 3 Serialization Site Manager (SSM), it gives manufacturers a practical way to manage serialized products on manual or offline stations while preserving compliance, traceability, and integration with the wider serialization landscape.
As part of Vimachem’s open serialization platform, this serialization and aggregation software integrates seamlessly with various IT systems and third-party equipment. It is the ideal solution to ensure regulatory compliance even for manual reworking and low-speed production.

Why Manufacturers choose it
Not every packaging activity belongs to a high-speed automated line. Manufacturers still need compliant ways to handle exceptions, low-volume productions and post-batch changes. Manual stations are widely positioned across the market for these use cases, especially where traceability must be preserved without disrupting the main line.
A practical fit for small-batch manufacturing
As pharma shifts toward smaller batches, more specialized products, and more flexible packaging operations, manual and semi-manual solutions become more relevant. That is especially true for CDMOs, regional manufacturers, and sites serving many markets with different serialization expectations.
MSAM Use Cases
Manual Serialization
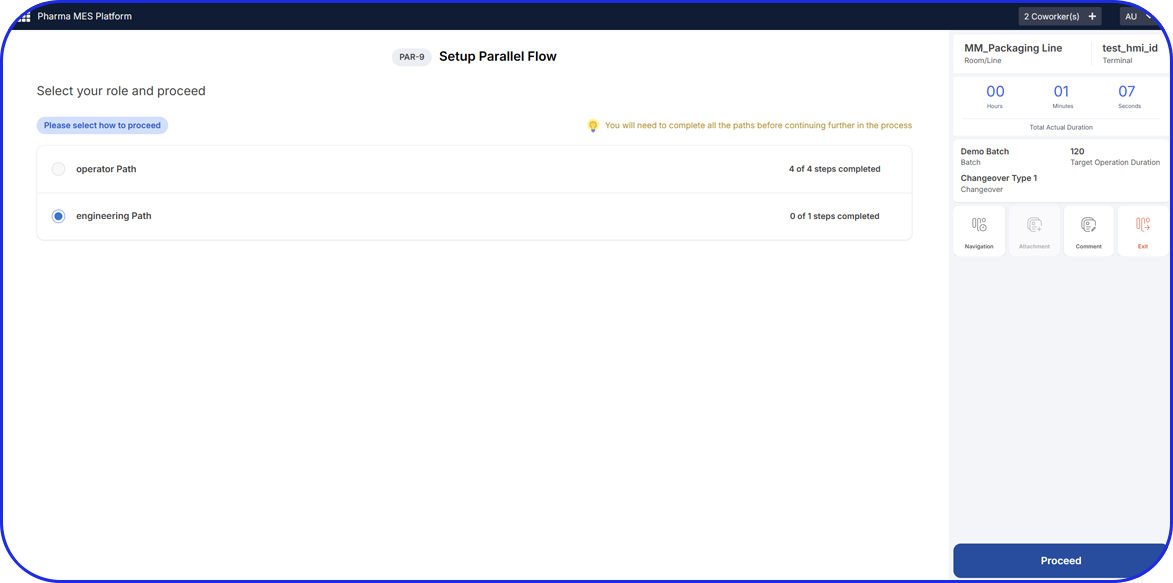
For products that need serialization outside a fully automated packaging line, MAM provides a controlled operator station approach using scanner, printer, and PC-based software. This is the right story for low-volume orders or sites that need flexible serialized capacity without redesigning the whole packaging flow.
Manual Aggregation
Aggregate serialized units into higher packaging levels and create parent-child relationships across up to three packaging levels. This supports full traceability from unit to bundle, shipper, or pallet and is especially relevant when manufacturers need compliant aggregation on manual or offline lines.
Manual Reworks
Handle sampling, damaged product, verification, relabeling, decommissioning, and post-batch corrections without sending product back to the original packaging line.
Benefits of Manual Serialization and Aggregation Software
Faster handling of returns, recalls, and exceptions
When serialized products need to be sampled, verified, reworked, or returned, pharmaceutical companies need a controlled way to intervene without sending material back to the packaging line. Vimachem’s Manual Serialization and Aggregation Module provides a compliant manual station for exception handling, helping manufacturers preserve traceability and respond faster to day-to-day warehouse or shopfloor issues. Connected to Vimachem Level 3 SSM, it allows these activities to be managed as part of the broader site serialization landscape rather than through disconnected manual workarounds.
Ready for emerging-market compliance needs
For manufacturers serving emerging and cost-aware markets, flexibility matters as much as compliance. A manual serialization and aggregation station provides a practical route to market readiness without the high investment and complexity of adding a dedicated automated packaging line. This is especially relevant for markets such as Egypt and Algeria, where serialization and traceability continue to advance as part of the sector’s broader digitization agenda. As an additional line within Vimachem Level 3 SSM, MSAM helps companies adapt to market-specific requirements with a more economical deployment model that still supports traceability and regulatory alignment.
Modular structure that fits the operation
MSAM gives manufacturers the flexibility to deploy exactly what the operation requires. It can be used as a serialization-only solution, an aggregation-only solution, or as a combined manual serialization and aggregation module for sites that need both capabilities in one station. That modular structure helps teams respond to different packaging setups and market requirements without overengineering the line. As part of Vimachem’s modular serialization landscape, MSAM supports a practical rollout path: start with the functionality you need today and expand as requirements evolve.
Frequently Asked Questions
What is aggregation in pharma?
Pharmaceutical aggregation refers to the process of linking individual pharmaceutical products to higher packaging levels (such as bundles, cases, or pallets) for traceability and compliance. It’s a critical step in the pharmaceutical supply chain, ensuring that products can be tracked from the smallest package to the final shipment. This process helps prevent counterfeiting and supports regulatory compliance in the pharma aggregation process.
How does pharmaceutical aggregation work?
Pharmaceutical aggregation works by assigning unique identifiers to individual packaging levels and linking them together. Using aggregation software, this data is then consolidated into higher packaging levels, ensuring complete traceability across the entire supply chain. The system captures and stores information such as serial numbers, batch numbers, and expiration dates, making it easy to track the product's journey from manufacturing to distribution. For low-speed lines, manual aggregation can be used to aggregate up to three packaging levels efficiently.
What is the difference between aggregation and serialization?
While serialization refers to assigning a unique serial number to each individual product unit, aggregation involves linking these serialized units to higher levels of packaging. In other words, aggregation in pharmaceutical products groups serial numbers from individual units into higher-level packages (e.g., boxes or pallets). Both processes are essential in ensuring the traceability and authenticity of pharma aggregation, but serialization focuses on individual units, while aggregation ensures complete packaging traceability.
Ready to Augment your Shop Floor Operations?
Ready to Augment your Shop Floor Operations?
Get started with real-time manufacturing analytics today.