Product Tour
Accelerating Digital Transformation in Pharma
Benefits of Electronic Batch Records
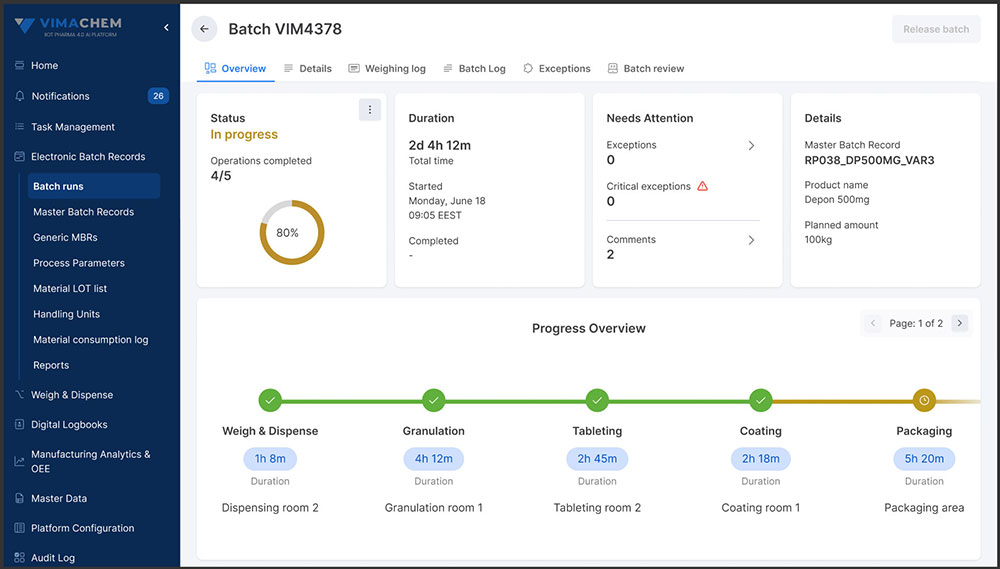
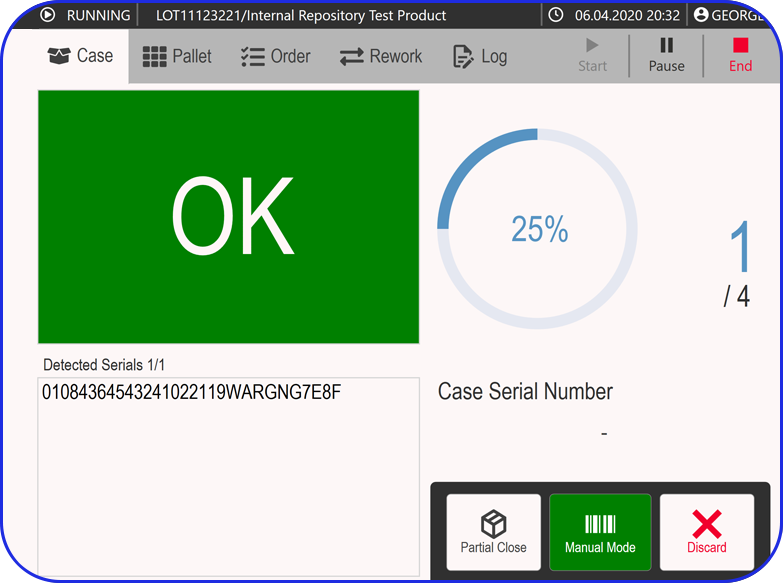
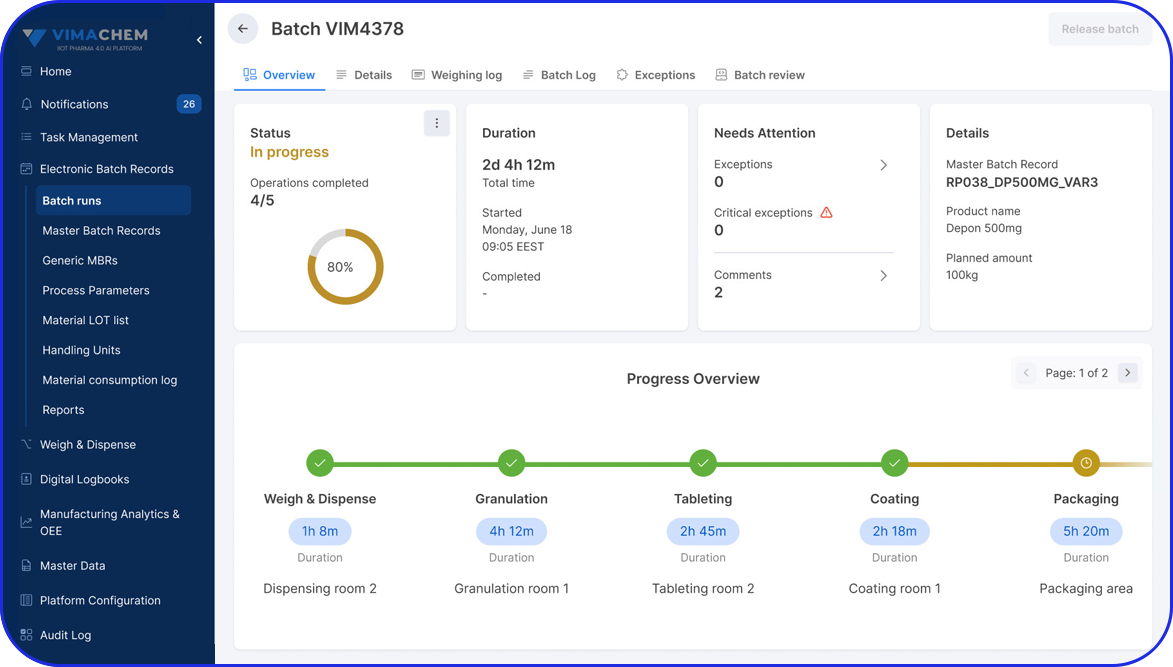
Faster, Smarter Batch Reviews
Manual batch reviews can be time-consuming and resource-intensive, often delaying product release. Vimachem’s review-by-exception approach surfaces critical deviations, allowing quality teams to focus their expertise where it’s needed most - accelerating approvals while upholding compliance and product integrity
Pre-Validated for Rapid Deployment
Implementation delays due to lengthy validation cycles can stall digital transformation and increase costs. Vimachem’s pre-validated solution includes a comprehensive, out-of-the-box validation package -reducing deployment timelines by at least two months while ensuring full compliance from the initial rollout.
Seamless System Integration
Disruptive system overhauls and poor interoperability can slow adoption and increase risk. Vimachem’s solution is built to integrate effortlessly with your existing ERP, MES, QMS, and lab systems -minimizing setup time and ensuring a smooth transition without added infrastructure investment.
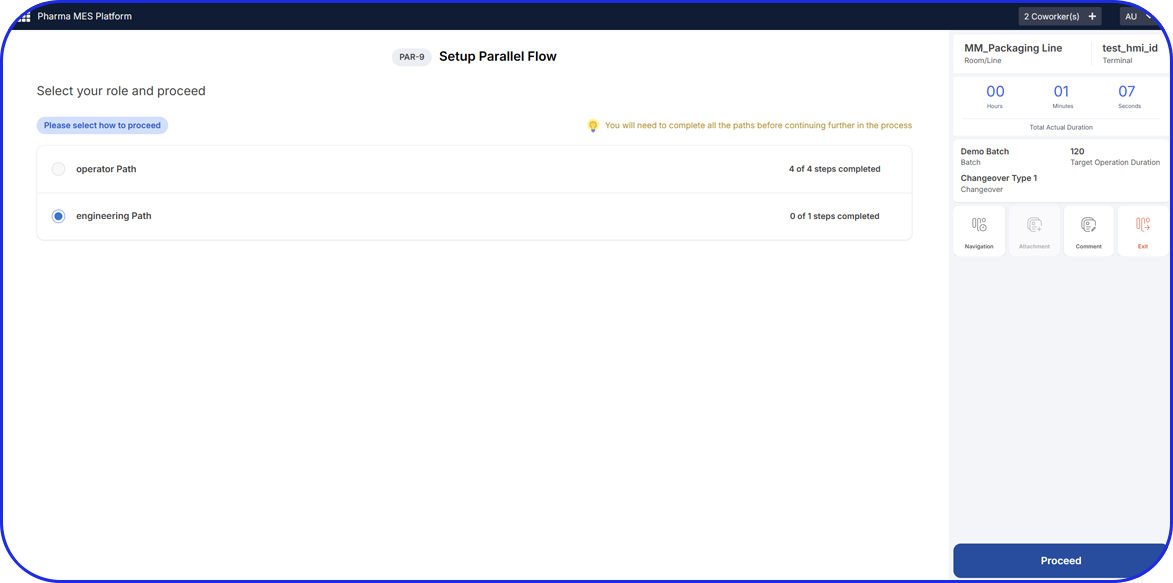
See Vimachem’s eBR in Action
Pharmaceutical Electronic Batch Records
Built for Manufacturing, Quality, and Compliance Teams in Pharma & Biopharma
Pharmaceutical and biopharmaceutical manufacturers face increasing pressure to ensure batch integrity, maintain compliance, and improve operational efficiency, all while reducing manual effort and review time. Vimachem’s Electronic Batch Records (eBR) solution addresses these challenges through automation, standardization, and real-time visibility – accelerating batch release and supporting regulatory compliance with minimal disruption.
Vimachem’s eBR solution empowers manufacturing and quality teams to digitize operations, maintain compliance, and accelerate batch release – delivering faster, smarter, and more resilient manufacturing across all sites.
Key Features of Our EBR Software
- A pre-validated solution with a complete IQ/OQ package, significantly reducing implementation time.
- Built-in compliance with FDA, GMP, and 21 CFR Part 11 for secure, validated, and auditable data and processes.
- A flexible framework that adapts easily to product changes, regulatory updates, and evolving processes.
- Seamless integration with IoT devices and smart sensors to improve data accuracy and enable real-time monitoring.
- Scalable architecture that supports multi-site operations and global manufacturing needs.
- Minimal training requirements thanks to an intuitive interface and streamlined workflows.
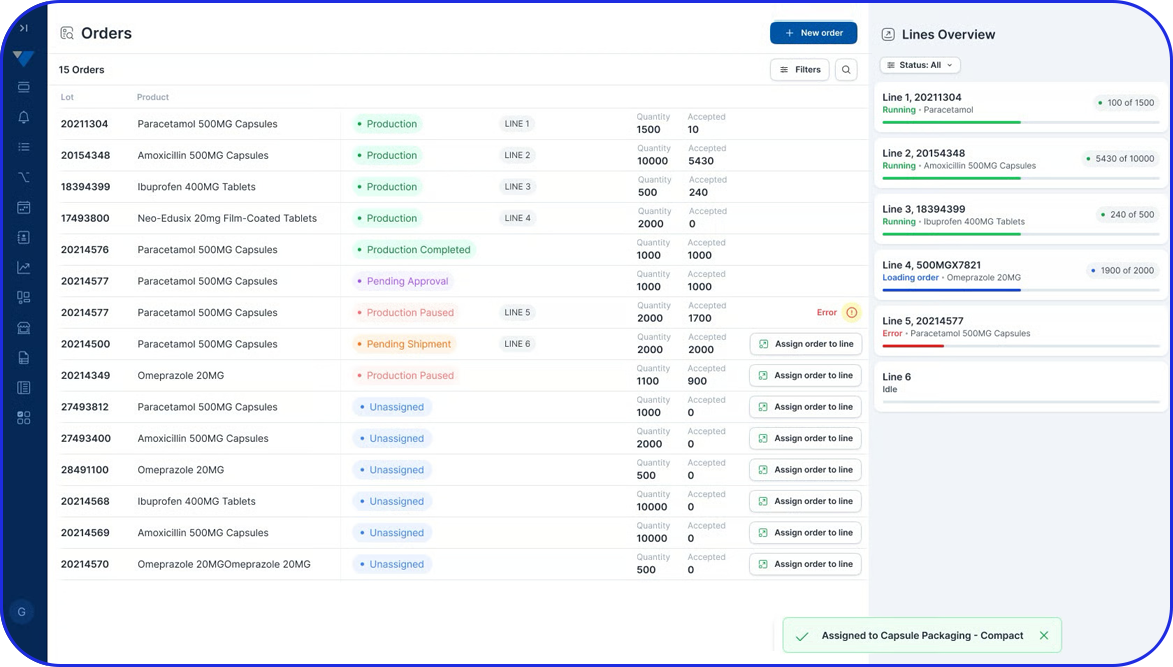
eBR Software Capabilities for Modern Manufacturing
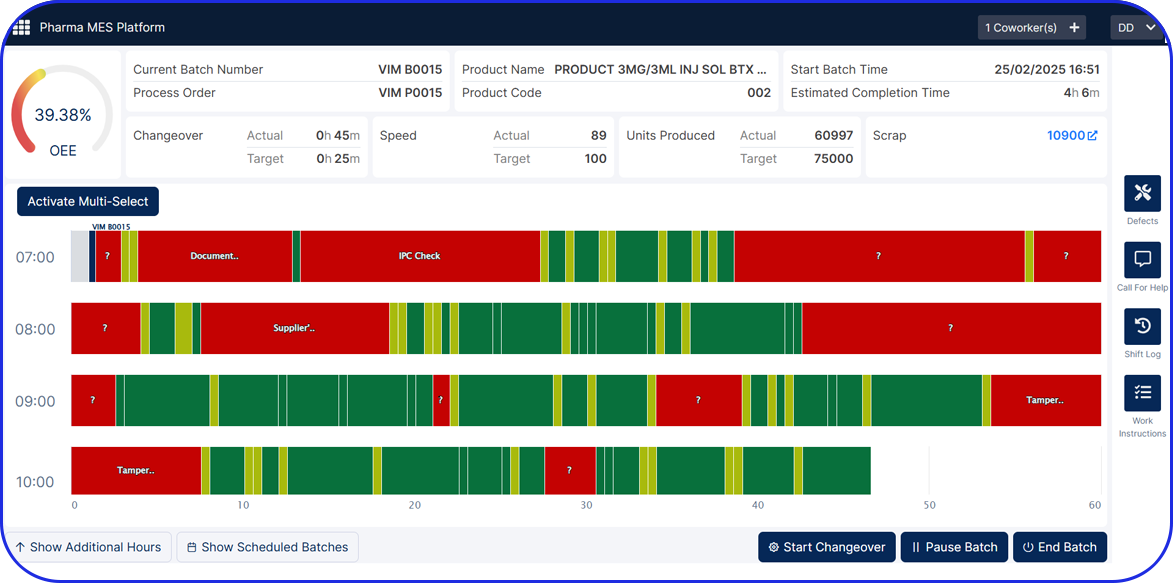
Automated Data Capture & Real-Time Visibility
Eliminate manual data entry with automated, real-time tracking of production activities - enhancing accuracy, reducing delays, and enabling data-driven decisions across your operations.
Faster, Right-First-Time Batch Reviews
Reduce QA review time through automated checks, exception handling, and right-first-time execution - minimizing rework, improving compliance, and speeding up batch release.
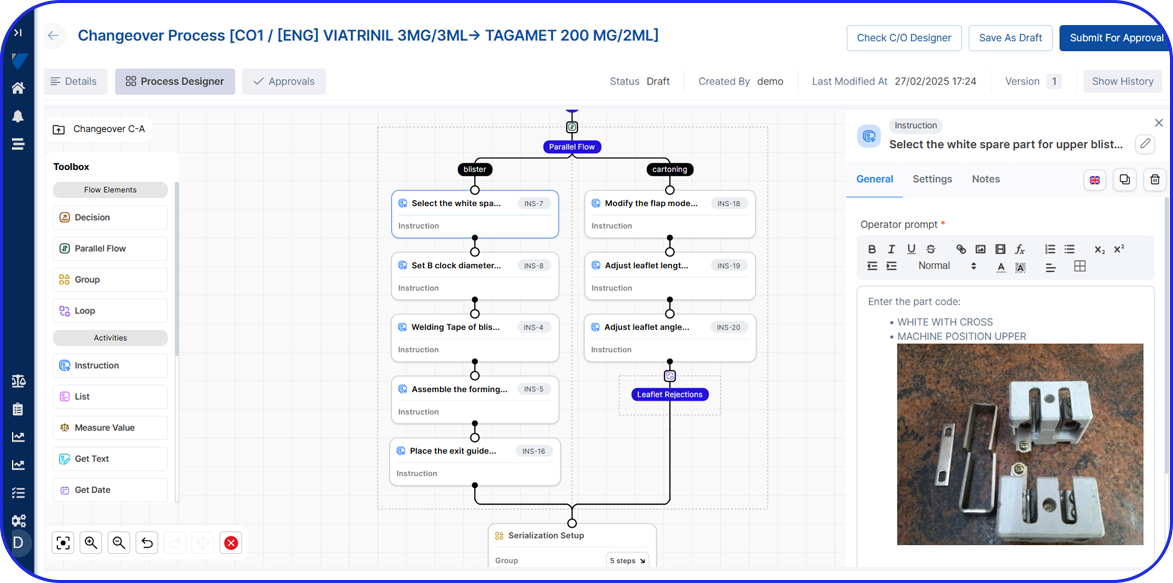
Configurable Workflows & Role-Based Access
Tailor workflows, approvals, and user permissions to match your specific processes - ensuring compliance with regulatory requirements while improving operational agility.
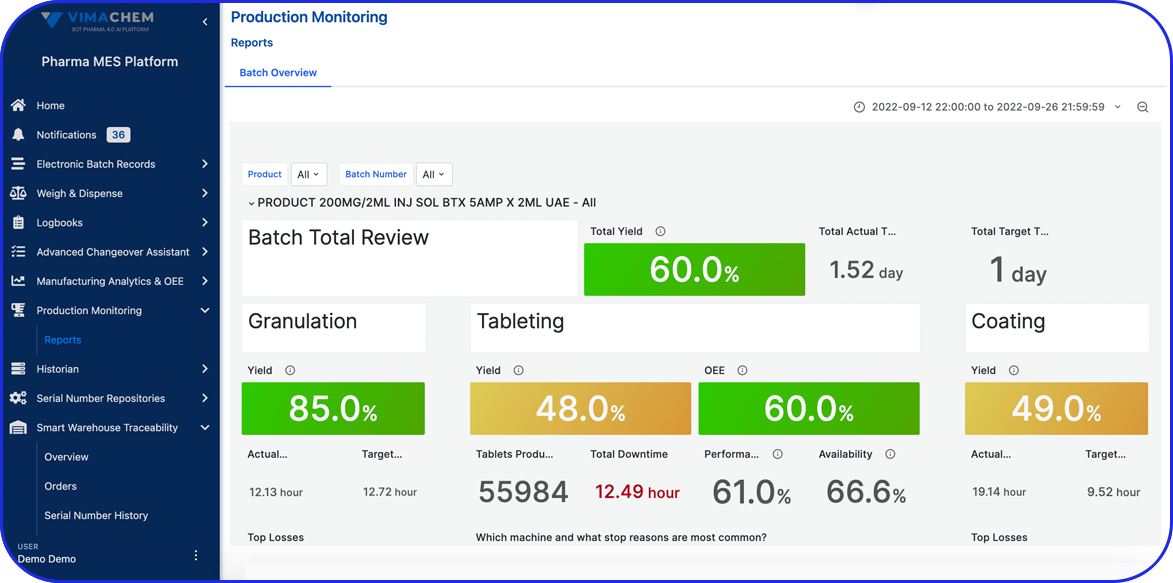
Advanced Reporting & Operational Dashboards
Generate detailed reports and visualize production trends with built-in dashboards - supporting continuous improvement, faster audits, and informed decision-making.
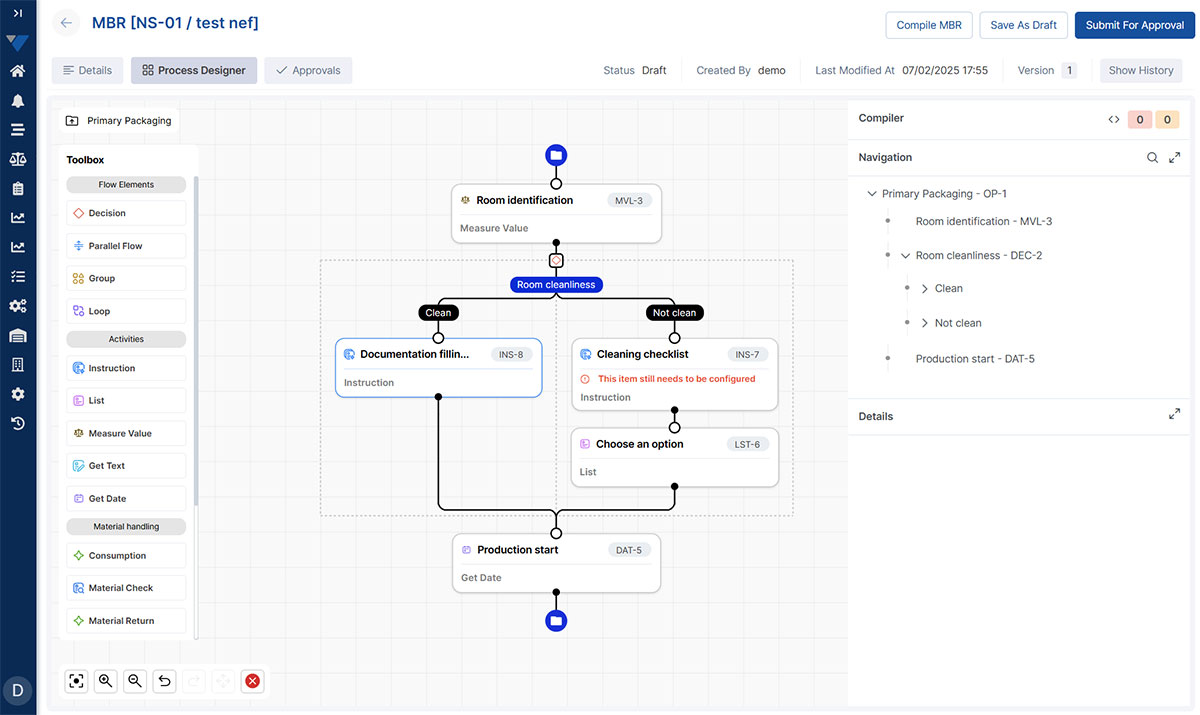
Scalable Recipe & Process Management
Adapt easily to new products, regulatory updates, or process changes with a flexible, low-maintenance recipe framework designed to grow with your operations.
Mobile-Friendly & Device
Agnostic
Access and manage production data securely from any device, anywhere - enabling real-time collaboration and responsiveness across teams and manufacturing sites.
Vimachem’s eBR has transformed our batch release, cutting review time by half while ensuring full regulatory compliance.
VP of Quality Operations, Global Pharmaceutical Manufacturing organization
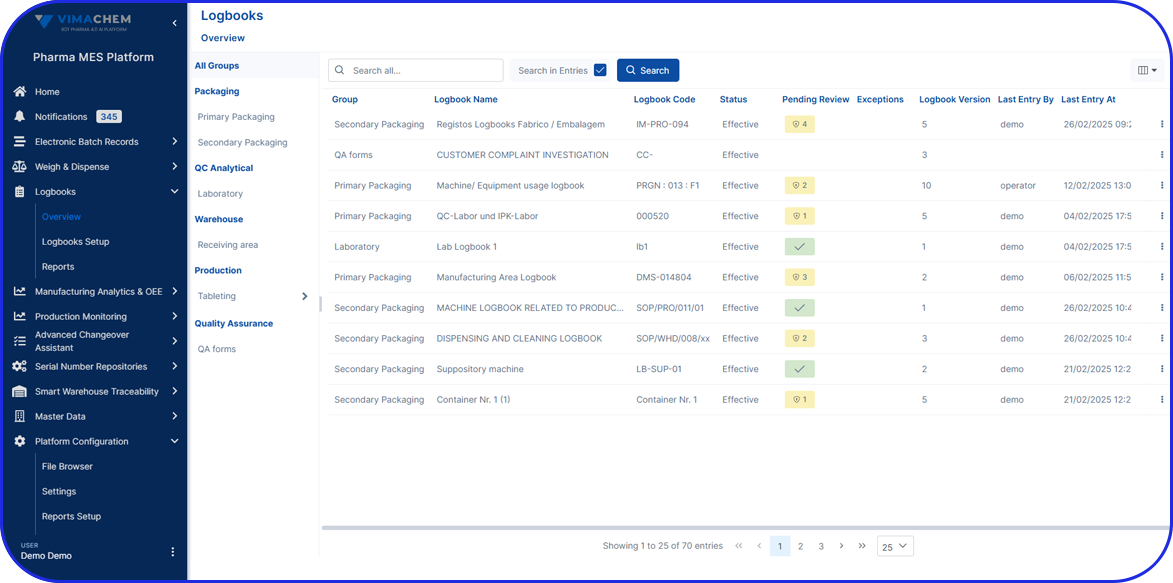
Validated EBR Software with GxP Compliance
User-Level Permissions & Security
Manage access with role-based permissions and Active Directory integration - enabling secure login, password policies, and simplified user administration with reduced IT overhead.
Verified Data Integrity
Ensure compliant, accurate data with built-in input validations, audit trails, and restricted access based on user roles - supporting data integrity at every stage of production.
Electronic Records & Signatures
Compliant with FDA 21 CFR Part 11 and EU GMP Annex 11, the system ensures secure, traceable, and auditable records with verified electronic signatures.
Complete Digital Records
Capture a full digital history of all shop floor activities, including device usage, material handling, and equipment logs - ensuring transparency and traceability.
Configurable Electronic Signatures
Enable efficient review and approval with electronic signatures embedded in workflow steps - ensuring compliance and consistency across all production stages.
Flexible Data Exports
Easily export data in XLSX and XML formats, manually or automatically - simplifying reporting, analysis, and system integrations.
Scalable EBR Solutions with Modular Add-Ons
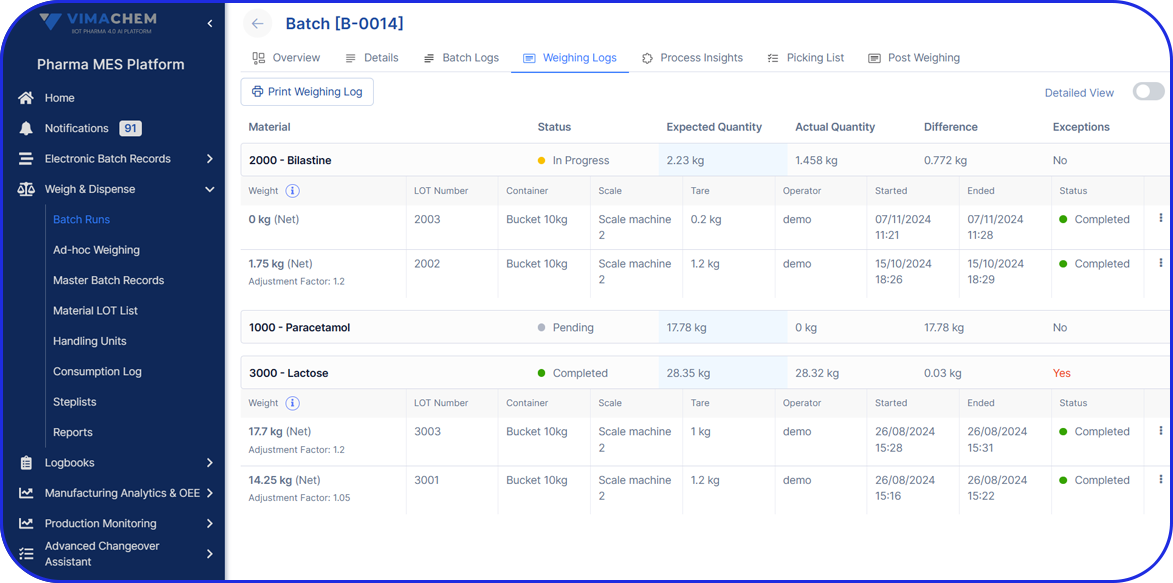
Vimachem’s eBR goes far beyond batch records – you can seamlessly integrate Manufacturing Analytics & OEE, and Weigh & Dispense to gain complete process visibility, operational excellence, and compliance across every stage of production.
Weigh & Dispense
Manufacturing Analytics & OEE
Contact us
FAQs on Electronic Batch Records
What is an Electronic Batch Record (EBR)?
An Electronic Batch Record (EBR) is a digital version of a paper batch record used in pharmaceutical and biopharmaceutical manufacturing. It captures, stores, and secures all production data in a compliant, auditable format — ensuring accuracy, traceability, and regulatory compliance.
What is the difference between an MBR (Master Batch Record) and an EBR?
A Master Batch Record (MBR) defines the recipe and instructions for manufacturing a product, while an Electronic Batch Record (EBR) documents the actual execution of each batch. Together, they ensure consistency, compliance, and quality in production.
What does EBR software do in pharmaceutical manufacturing?
EBR software automates data capture, enforces workflows, and provides real-time visibility into production. It reduces manual effort, minimizes errors, accelerates batch release, and ensures compliance with GMP and FDA regulations.
How do Electronic Batch Records support GMP and GxP compliance?
EBR software enforces Good Manufacturing Practice (GMP) and GxP compliance through audit trails, electronic signatures, role-based access, and built-in data integrity checks ensuring every batch meets regulatory requirements.
Is Vimachem’s EBR software validated and FDA 21 CFR Part 11 compliant?
Yes. Vimachem’s EBR is a validated solution with a complete IQ/OQ package. It complies with FDA 21 CFR Part 11, EU Annex 11, and GMP standards providing secure, traceable, and auditable records from day one.
What are the benefits of using Electronic Batch Records in pharma and biopharma?
The key benefits include faster batch release, reduced QA review time, improved data integrity, seamless regulatory compliance, lower error rates, and real-time visibility across manufacturing operations.
How does EBR software help accelerate batch release and reviews?
With review-by-exception functionality, EBR software flags only deviations and critical issues for QA teams. This reduces manual review effort, shortens approval timelines, and enables faster, right-first-time batch release.
Can EBR integrate with ERP, MES, QMS, and LIMS systems?
Yes. Vimachem’s EBR integrates seamlessly with ERP, MES, QMS, and LIMS systems. This ensures smooth data exchange, eliminates silos, and supports a connected digital manufacturing ecosystem.
What is CSV (Computer System Validation) for Electronic Batch Records?
CSV is the process of verifying and documenting that EBR software functions as intended and meets regulatory requirements. A validated EBR system ensures accuracy, reliability, and compliance throughout its lifecycle.
How long does it take to implement validated EBR software?
Implementation timelines vary, but Vimachem’s pre-validated EBR solution reduces deployment by at least two months. Its out-of-the-box validation package ensures rapid rollout with minimal disruption.
Ready to Augment your Shop Floor Operations?
Ready to Augment your Shop Floor Operations?
Get started with real-time manufacturing analytics today.