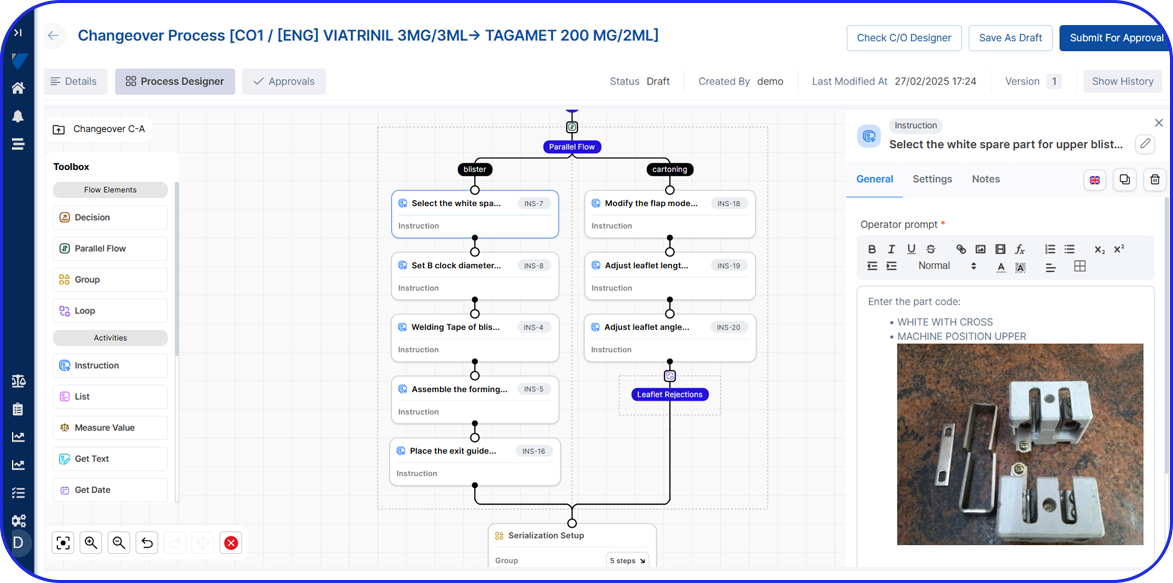
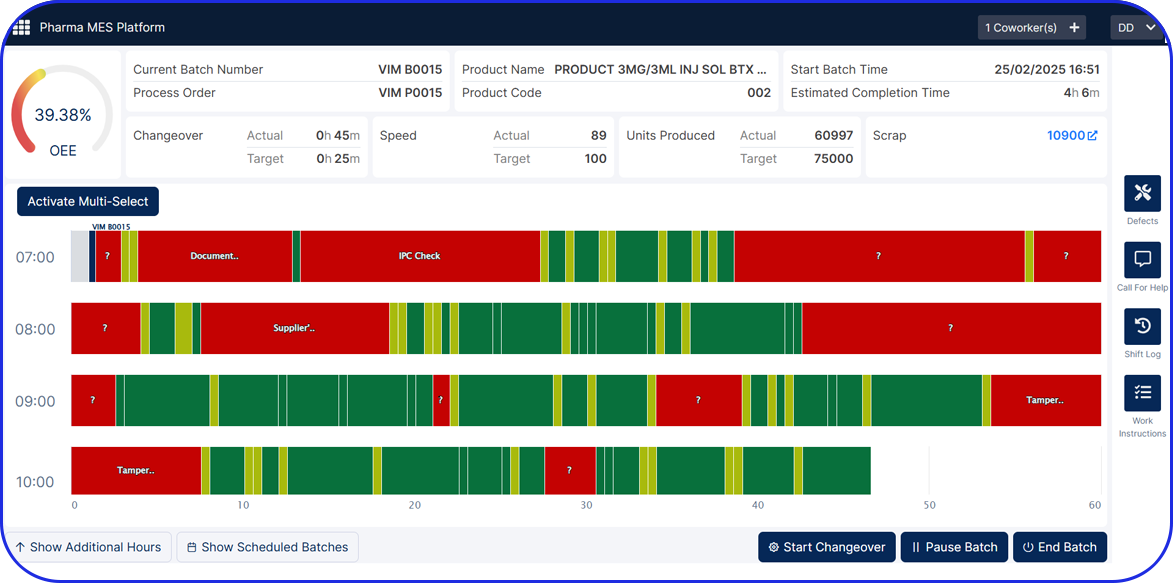
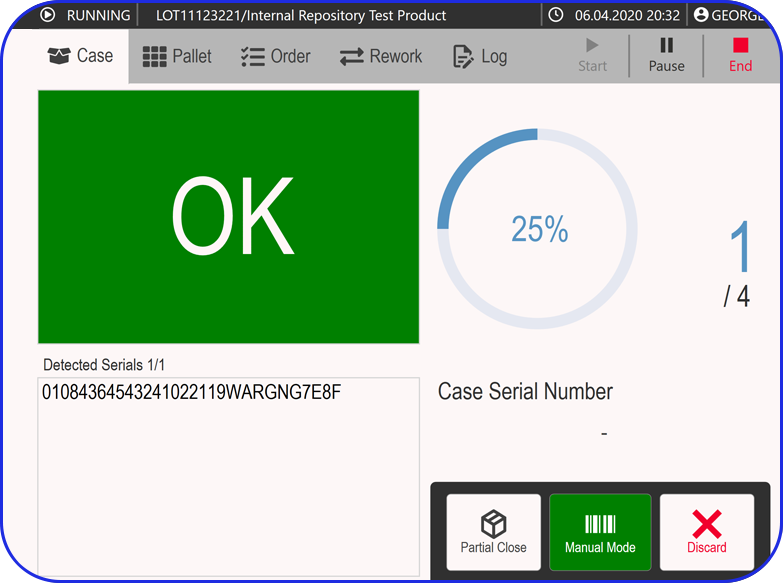
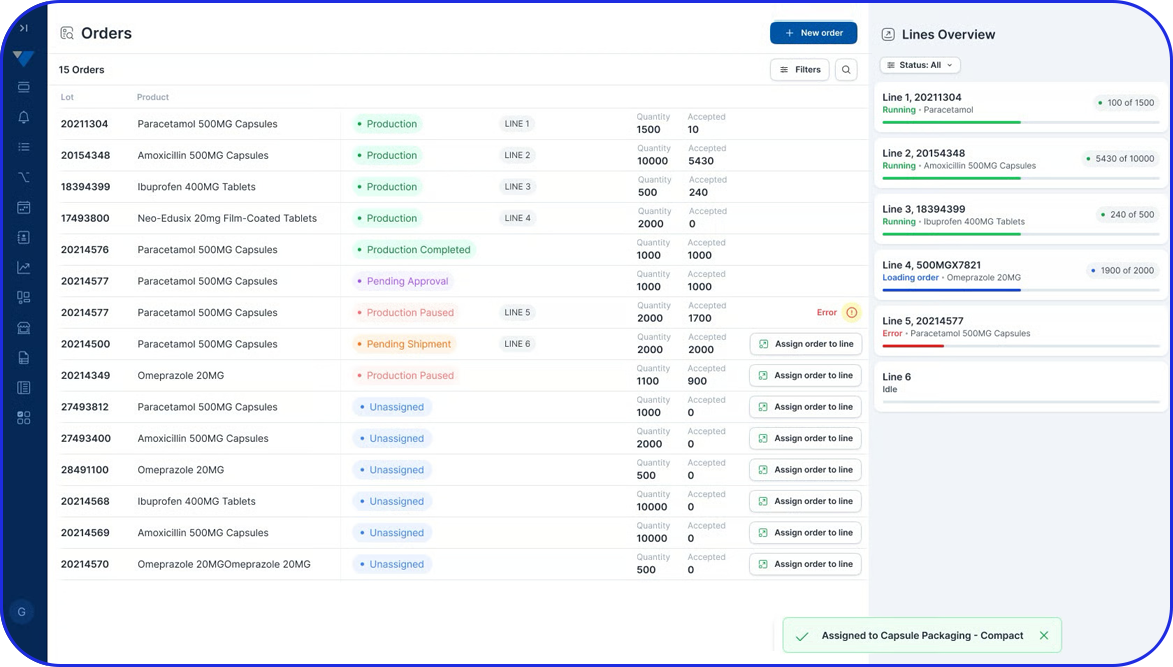
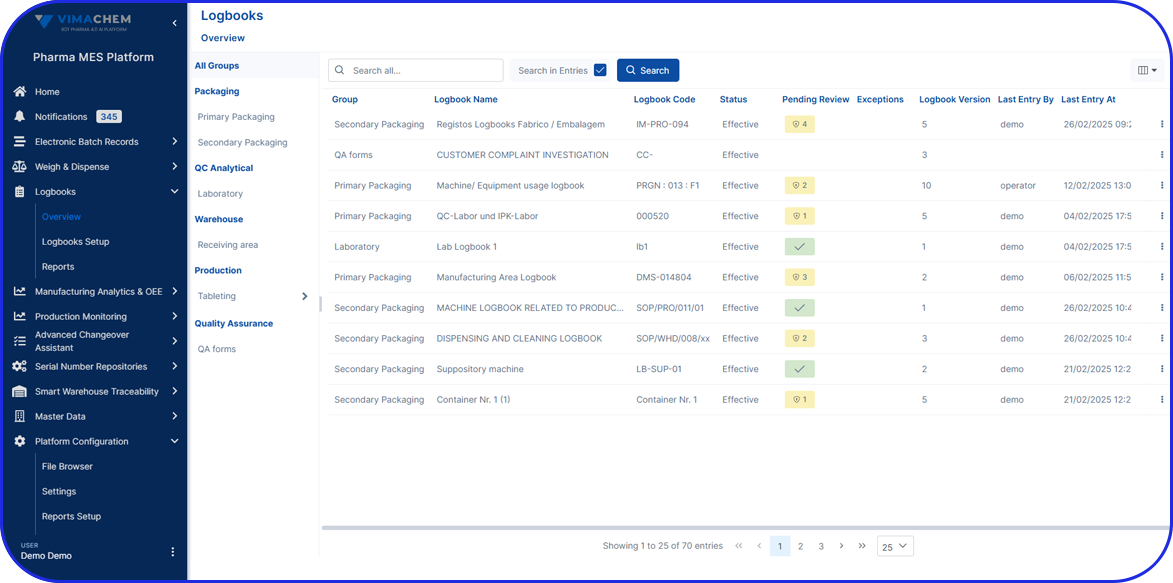
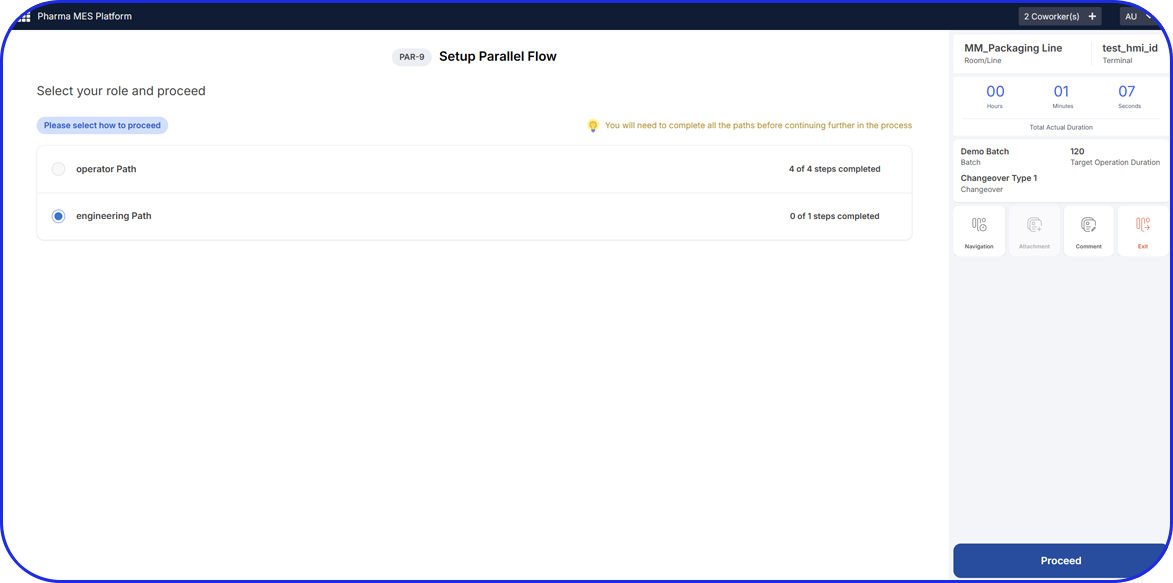
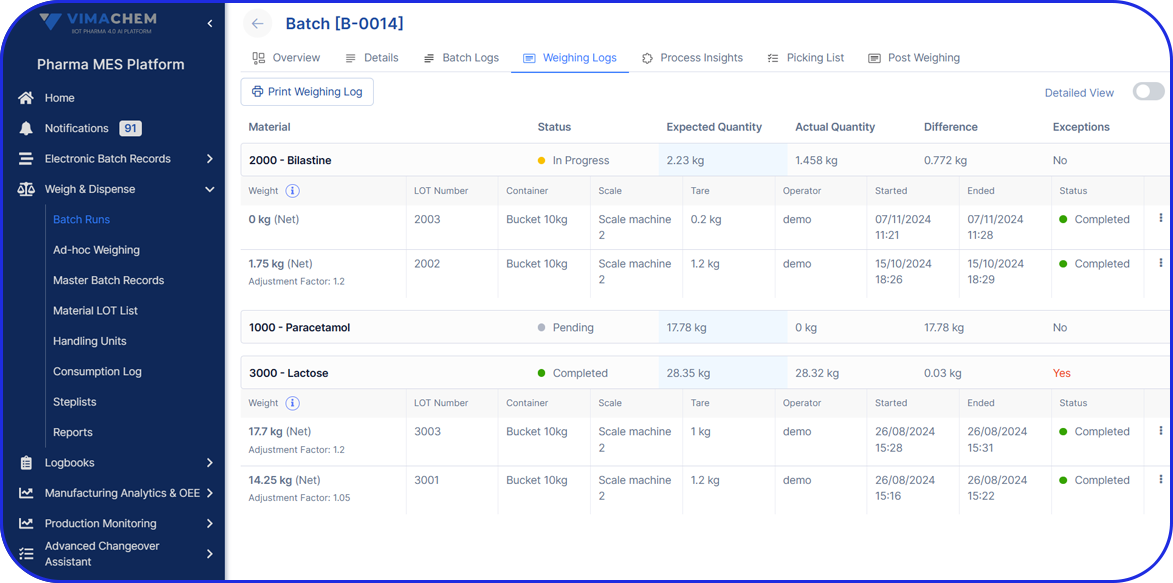
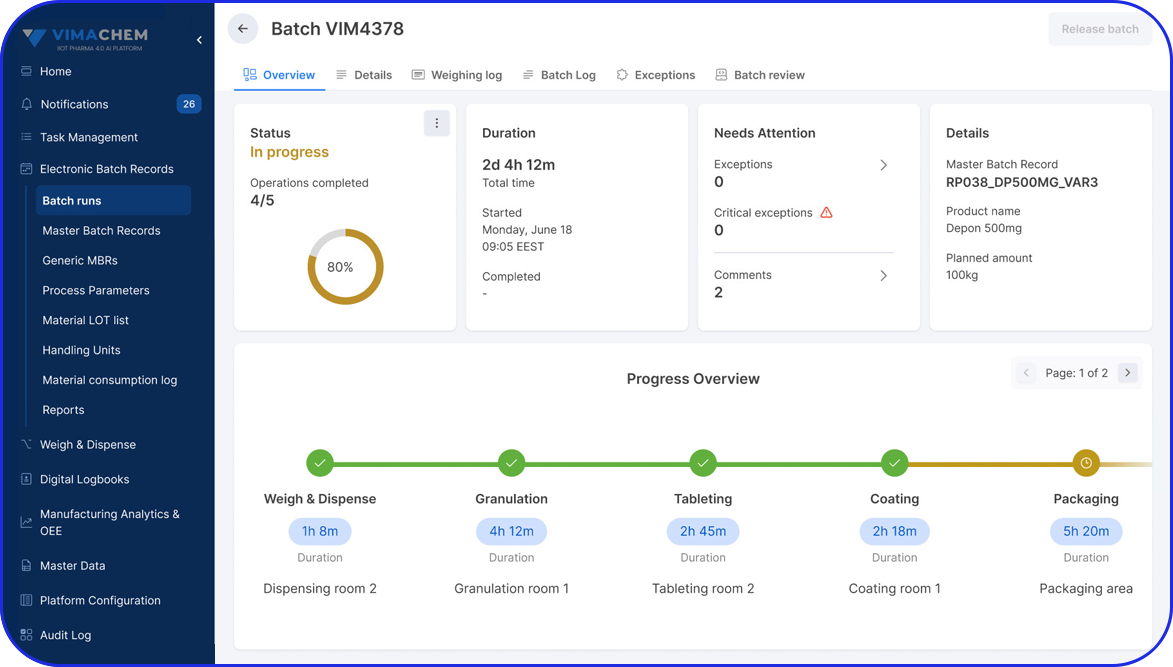
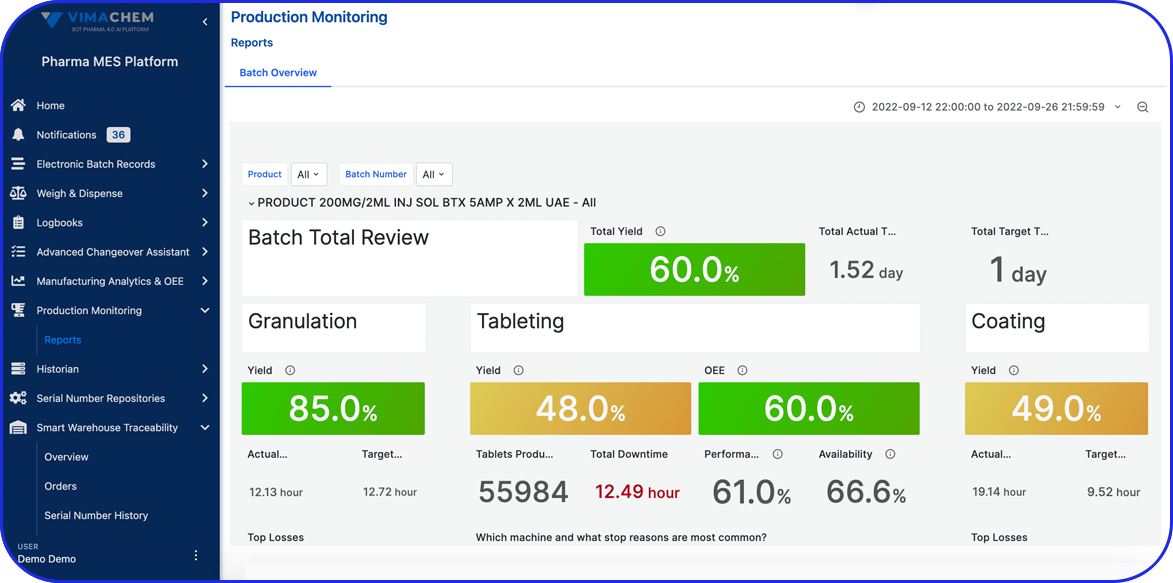
Accelerating Value Through Digitization: A CDMO Case Study on Parallel ERP & eBR Implementation Accelerating Value Through Digitization: A CDMO Case Study on Parallel ERP & eBR Implementation Emma Hanley Pharma Digitization Manager Linkedin ERP & EBR Go live readiness: Up to 50% faster Revenue Realization: Up to 18 months earlier Modern pharma manufacturers can […]
Egypt Pharma Track & Trace: What’s Coming in 2026 and How Vimachem Can Help You Comply Egypt Pharma Track & Trace: What’s Coming in 2026 and How Vimachem Can Help You Comply Saud Khan Senior IT Serialization Consultant Linkedin Egypt is moving quickly toward full pharmaceutical traceability with key deadlines in February and August 2026. […]
Algeria Pharma Serialization: What’s coming in 2027 and how Vimachem can help you Algeria Pharma Serialization: What’s coming in 2027 and how Vimachem can help you Saud Khan Senior IT Serialization Consultant Linkedin Algeria is officially introducing pharmaceutical serialization and aggregation requirements in January 2027. While technical details are still being finalized by the National Agency for […]
// Configurable Not Custom Why Pharma Manufacturing Needs a No-Code MES Purpose-Built for Pharma // Configurable Not Custom Why Pharma Manufacturing Needs a No-Code MES Purpose-Built for Pharma Emma Hanley Senior Product Marketing Manager Linkedin Pharmaceutical manufacturing is under increasing pressure to digitalize faster, smarter, and without compromising compliance. Regulatory scrutiny is intensifying. Global supply […]
Digital-By-Design Pharma: Why the Next Era of Pharma Manufacturing Requires MES as Core CAPEX – not a Side Project Digital-By-Design Pharma: Why the Next Era of Pharma Manufacturing Requires MES as Core CAPEX – not a Side Project Emma Hanley Pharma Digital Transformation Consultant Linkedin Reflections from the ISPE Ireland Annual Conference 2025 -“Pioneering Tomorrow: […]
Digitizing Review and Release: Smarter Workflows, Faster Reviews, Stronger Compliance with Vimachem eBR Digitizing Review and Release: Smarter Workflows, Faster Reviews, Stronger Compliance with Vimachem eBR Emma Hanley Senior Product Marketing Manager In pharma manufacturing, batch review and release can be a bottleneck. Paper-based processes slow production, introduce human error, and delay delivery, while regulators […]
Why GMP-Critical Processes Don’t Belong in ERP: W&D, Logbooks, and Electronic Batch Records (eBR) Why GMP-Critical Processes Don’t Belong in ERP: W&D, Logbooks, and Electronic Batch Records (eBR) Emma Hanley Senior Product Marketing Manager Introduction ERP systems are powerful. They centralize planning, optimize resources, and give organizations visibility across the enterprise. It’s no surprise many […]
ERP Can’t Weigh Your Batches: The Case for MES-Native Weigh & Dispense ERP Can’t Weigh Your Batches: The Case for MES-Native Weigh & Dispense Emma Hanley Senior Product Marketing Manager Introduction In pharmaceutical manufacturing, precision is everything. Every gram matter, every action must be traceable, and every record must withstand regulatory scrutiny. Yet many companies […]
Should you integrate Electronic Logbooks and OEE? Should you integrate Electronic Logbooks and OEE? Łukasz Iskra Business Development Director Linkedin As someone who regularly supports pharma and biopharma companies – especially CDMOs, CPOs – on their digital transformation journey, I often get the same question:“Should we integrate Electronic Logbooks with OEE data?”My answer is: absolutely […]
Unlocking Pharmaceutical Excellence: The FDA Quality Maturity Model Unlocking Pharmaceutical Excellence: The FDA Quality Maturity Model Nefeli Stylla QA & CSV Specialist Linkedin Are manual processes slowing down your compliance efforts? Is it time for the pharmaceutical industry to enhance operations and shift towards digital transformation? FDA’s Quality Maturity Model (QMM) can serve as your […]