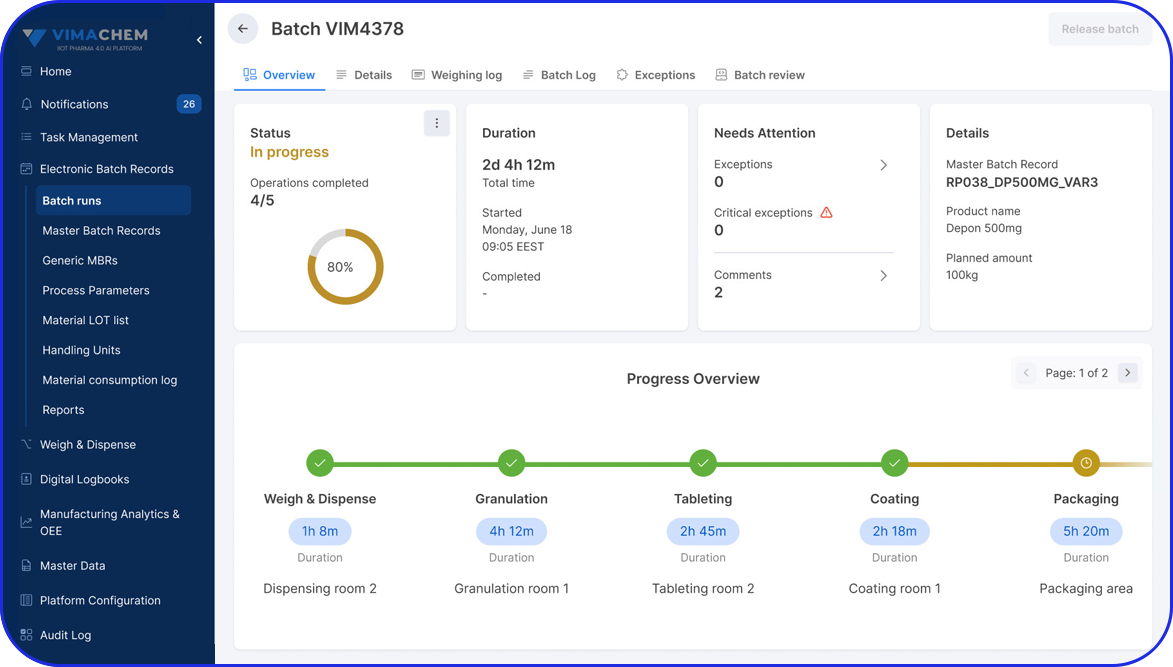
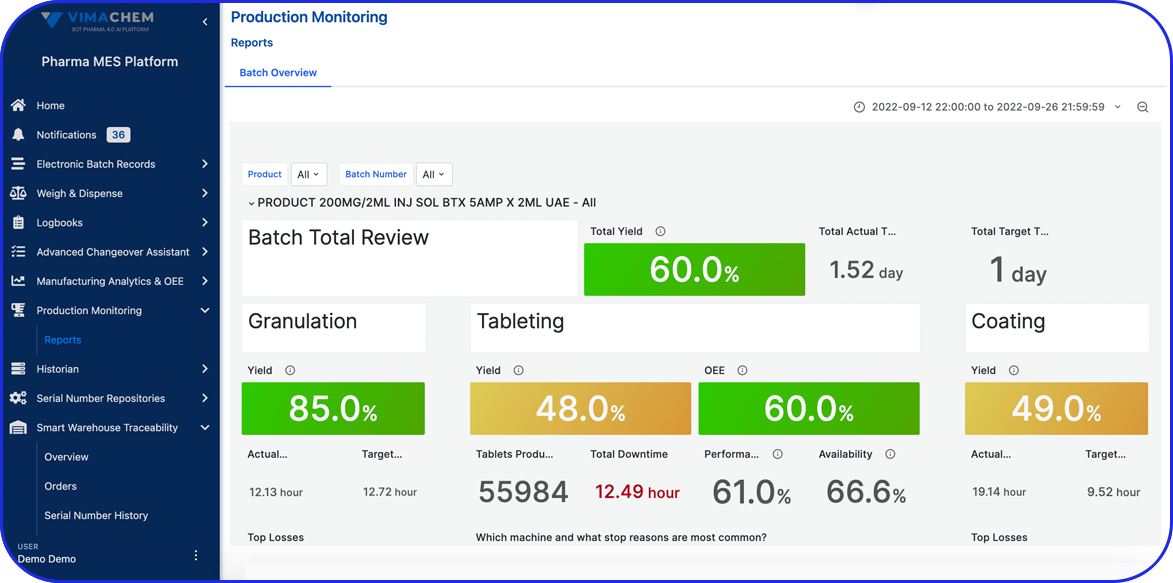
Algeria Pharma Serialization: What's coming in 2027 and how Vimachem can help you
Algeria Pharma Serialization: What's coming in 2027 and how Vimachem can help you

Saud Khan
Senior IT Serialization Consultant
Algeria is officially introducing pharmaceutical serialization and aggregation requirements in January 2027. While technical details are still being finalized by the National Agency for Pharmaceutical Products (ANPP), the direction is clear: serialized 2D coding, aggregation, and national reporting will become mandatory on January 1, 2027. Market Authorization Holders (MAHs), manufacturers, CMOs, and logistics partners supplying Algeria should already be preparing.
Algeria’s choice mirrors an international movement toward product traceability, supply chain openness and measures against counterfeiting. Serialization is no longer confined to the EU, US or major export destinations. It is emerging as a standard throughout Africa, the Middle East and Asia. Although technical criteria may still develop the path is evident: businesses delivering to Algeria should start getting ready not just at the packaging line stage but also, throughout storage, warehousing, and reporting systems.
For serialization at the unit level, the standard method is to use a GS1 DataMatrix on the unit or carton which encodes essential identifiers including the GTIN, batch number, batch expiration date and an exclusive serial number. Moreover, the establishment of parent–child relationships via aggregation is anticipated, usually employing GS1 logistics identifiers, like SSCCs.
To make traceability operational, companies should also prepare for a serialization repository and reporting hub to be able to exchange serialization data with supply chain partners through standardized methods. The ANPP has expressed plans to implement a reporting ecosystem further down the line in 2027.
Who should act now?
The scope impacts the broader supply chain such as MAHs importing into Algeria, local manufacturers, CMOs packaging for Algeria as well as 3PLs and warehouses handling serialized and aggregated stock. The practical reality shows: once aggregation becomes expected, warehouse operations must be serialization-aware (receiving, put-away, picking, shipping, returns), not just packaging lines.
Why early preparation matters
Serialization regulations touch far more than packaging lines. They affect IT architecture and data governance, warehouse operations, quality systems and regulatory reporting processes. Organizations that succeed typically approach serialization as a cross-functional program, not a single compliance task. The question is no longer whether companies should prepare, but how they do so efficiently.
What does good preparation look like?
Based on global serialization rollouts, good preparation for Algeria involves readiness across three interconnected areas:
- Packaging Line Readiness
At the physical packaging level, companies must ensure that production lines are technically capable of serialization and aggregation. Without stable, well-trained line operations, even the best IT systems will struggle. - Serialization Management Readiness
Beyond the line, companies need centralized control over serial numbers and product data. Key elements include a clear master data governance and integration readiness with external partners. - Warehouse and Distribution Readiness
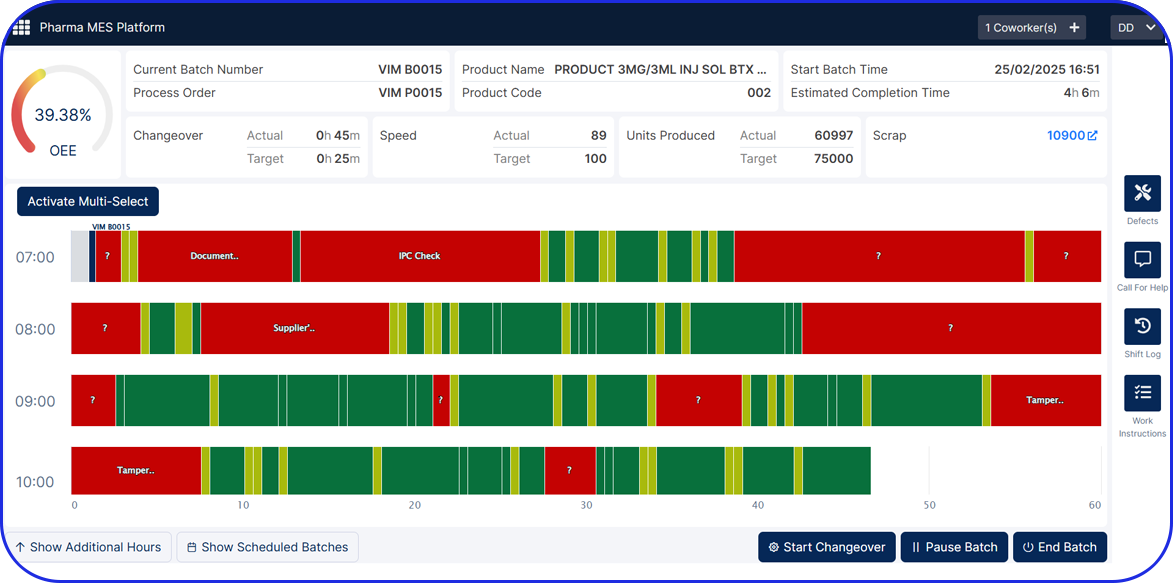
Serialization does not end when products leave the packaging line. Warehouses and distribution centers must be able to handle serialized and aggregated products without breaking data integrity. This is where serialization meets real-world logistics and where smart warehousing capabilities become critical.
Why choose Vimachem to support Algeria compliance?
At Vimachem, our key differentiator is not just compliance but flexibility, scalability, and operational excellence.
Direct Connectivity to the Algerian Reporting Repository
A central element of Algeria’s upcoming regulation is national serialization reporting. Vimachem’s primary offering enables MAHs to connect directly to the Algerian reporting repository through our Level 3 and 4 Serialization Site Manager. Vimachem manages the complete serial number lifecycle, event reporting, and data exchange required for regulatory submission.
Modular, Flexible, and Line-Agnostic
Vimachem’s platform is modular by design, enabling companies to deploy only what they need, when they need it. The Vimachem Serialization Site Manager Level 3 and 4 solution is L2-agnostic, giving our customers the flexibility to select the most appropriate Level 2 solution for each packaging line from high-speed automated lines to low-speed or semi-automatic lines or even fully manual serialization and aggregation scenarios. This flexibility avoids overengineering and significantly reduces long-term cost and vendor lock-in. With 50+ customers globally using various L2s across the site, we have proven that flexibility and avoidance of vendor lock-in are the most secure and cost-efficient way to initiate and scale your pharmaceutical serialization operations.
Built for Zero Downtime Operations
Serialization should never compromise supply continuity. Vimachem’s solutions are designed to go-live with no production downtime, supporting parallel rollout, phased go-lives, and controlled validation strategies. This allows companies to remain fully operational while upgrading their serialization and reporting capabilities.
The Vimachem Serialization Modules
We provide pharmaceutical companies with modular and regulation-ready serialization solutions that support pharmaceutical companies across packaging, warehousing, and distribution. Our portfolio is designed to scale with evolving global requirements, including emerging markets such as Algeria:
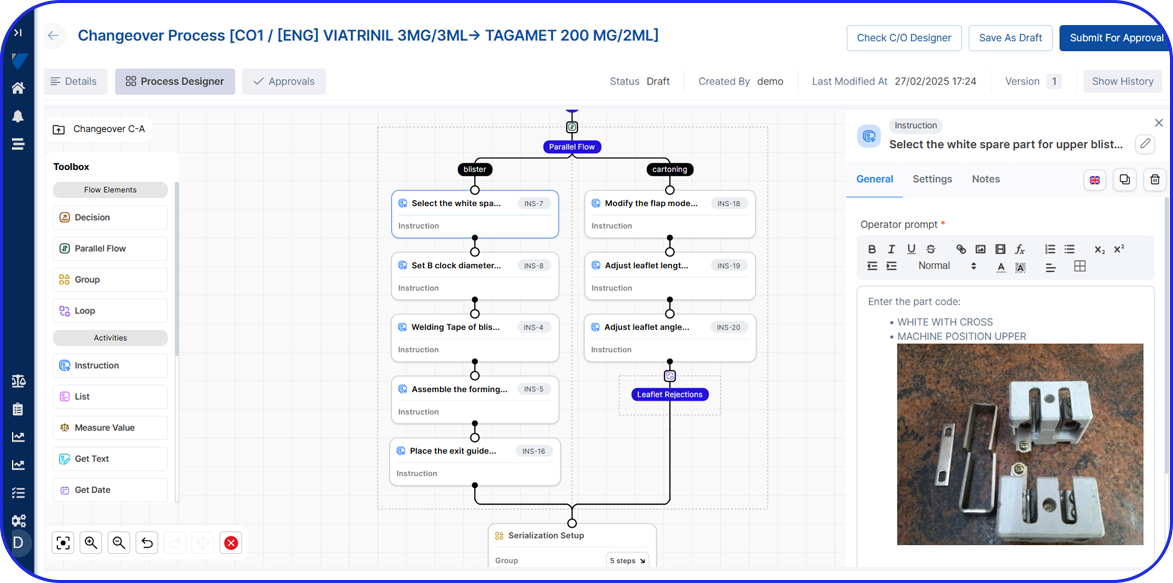
Vimachem Level 3&4 Serialization Site Manager (SSM)
Vimachem’s Site Manager (SSM) is a Level 3 and 4 serialization and aggregation solution that serves as the central hub between packaging lines (Level 2) and reporting systems (e.g EU Hub and/or Algerian Reporting Hub). It manages serial numbers, product and batch data, aggregation hierarchies, compliance reporting and remote line management while providing real-time visibility, analytics, and full regulatory compliance.
See the Product Tour
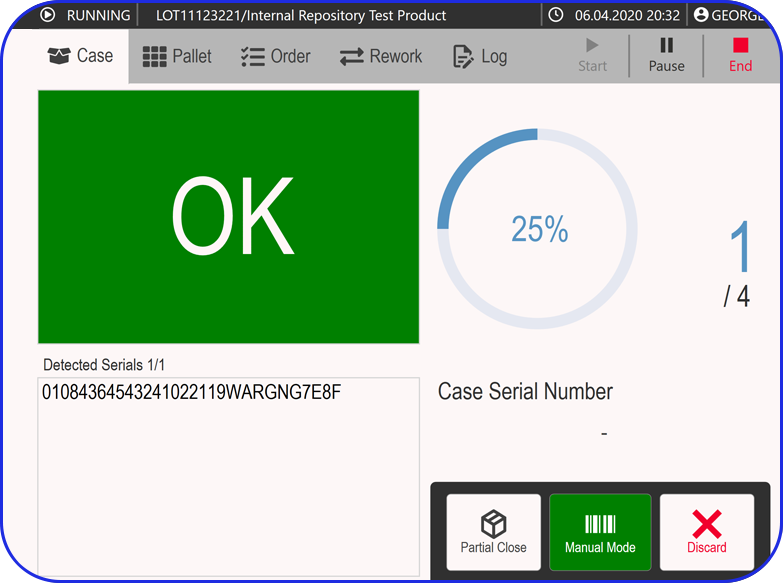
Vimachem Smart Warehouse Traceability (SWT)
Vimachem’s Smart Warehouse Traceability (SWT) is a dedicated warehousing traceability module built for pharmaceutical, biotech, and logistics operations that require serialized and aggregated product tracking including scan-driven receiving and shipping. SWT bridges the gap between ERP, WMS, printers, and serialization systems to enable real-time visibility and compliance across the warehouse lifecycle.
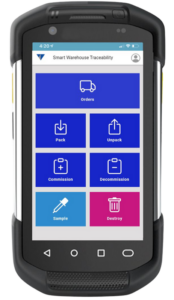
Vimachem Manual Serialization and Aggregation Modules (MSAM)
Our Manual Serialization and Aggregation module is a lightweight, operator-driven serialization and aggregation tool that allows human operators to enter or scan serial numbers manually. MSAM would bridge gaps where full line-level serialization and/or aggregation is not available, ensuring consistent traceability even in manual segments.
Looking Ahead to 2027
Serialization in Algeria is coming and preparation should start now. Successful compliance will depend on coordinated readiness across packaging, IT, warehousing, and quality functions. Companies that act early can avoid rushed implementation, reduce operational disruption and align serialization with broader digitalization goals. Those that wait risk facing higher costs, tighter timelines, and increased compliance pressure. With proven serialization and smart warehousing solutions, Vimachem is positioned to help pharmaceutical companies navigate Algeria’s requirements with confidence, ensuring compliance while strengthening supply chain visibility and control.