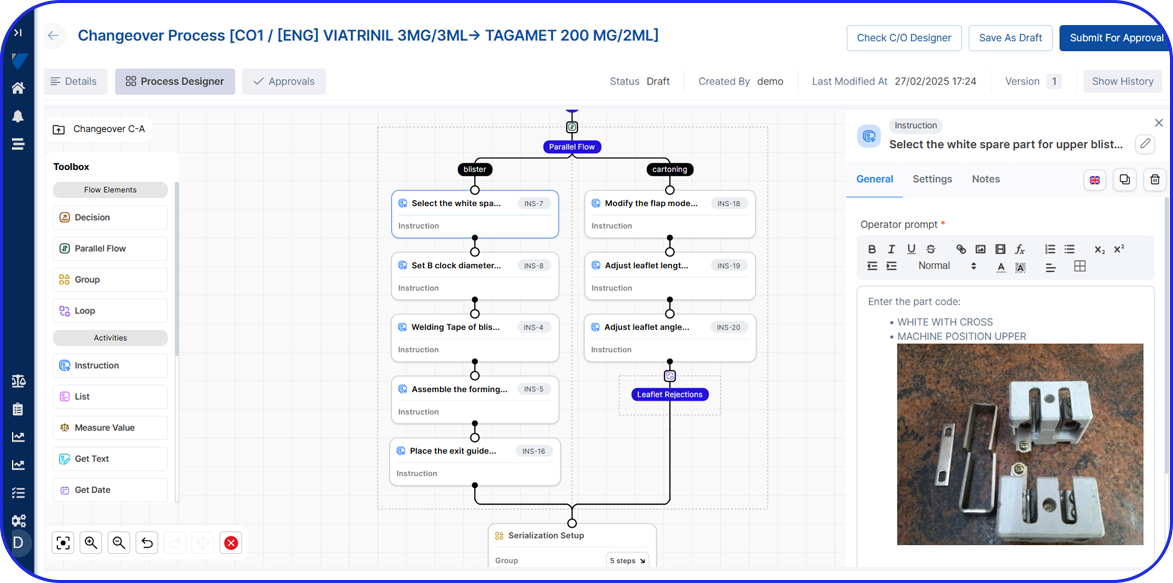
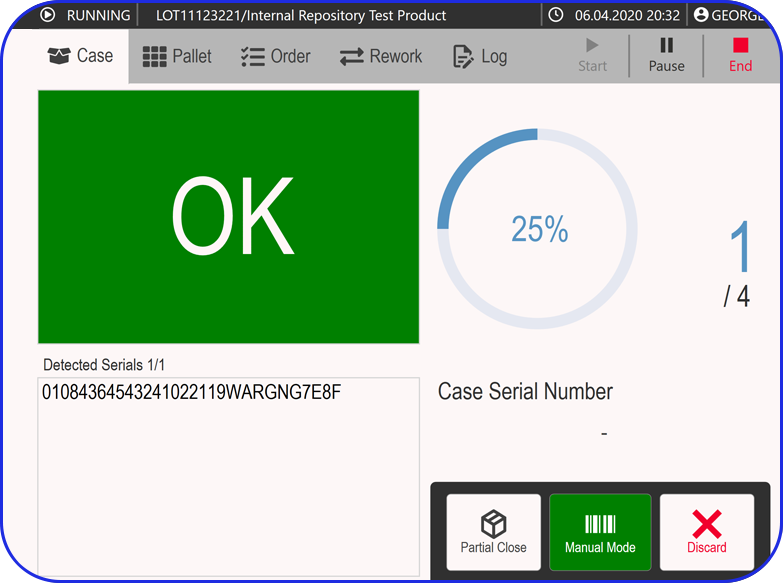
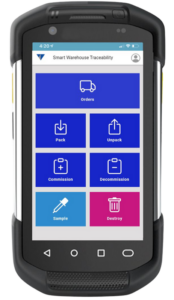
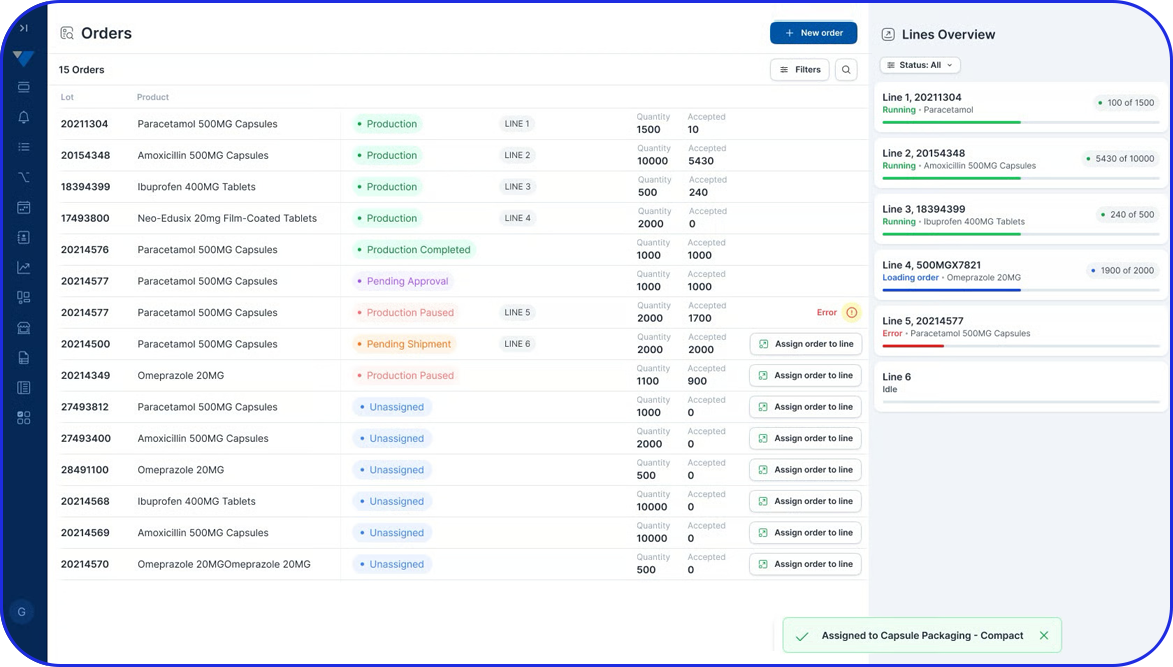
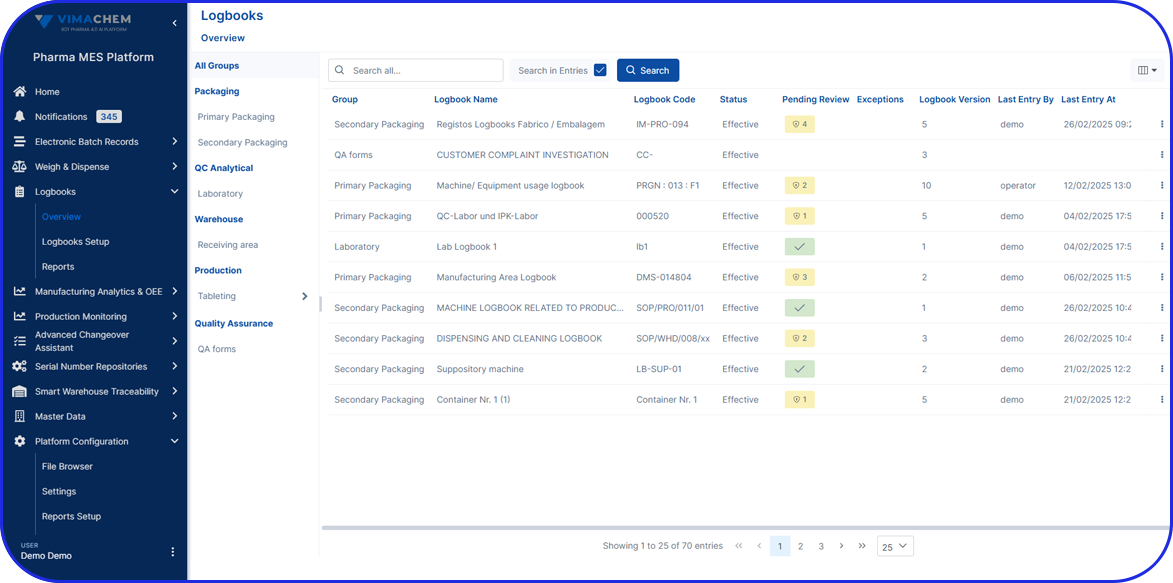
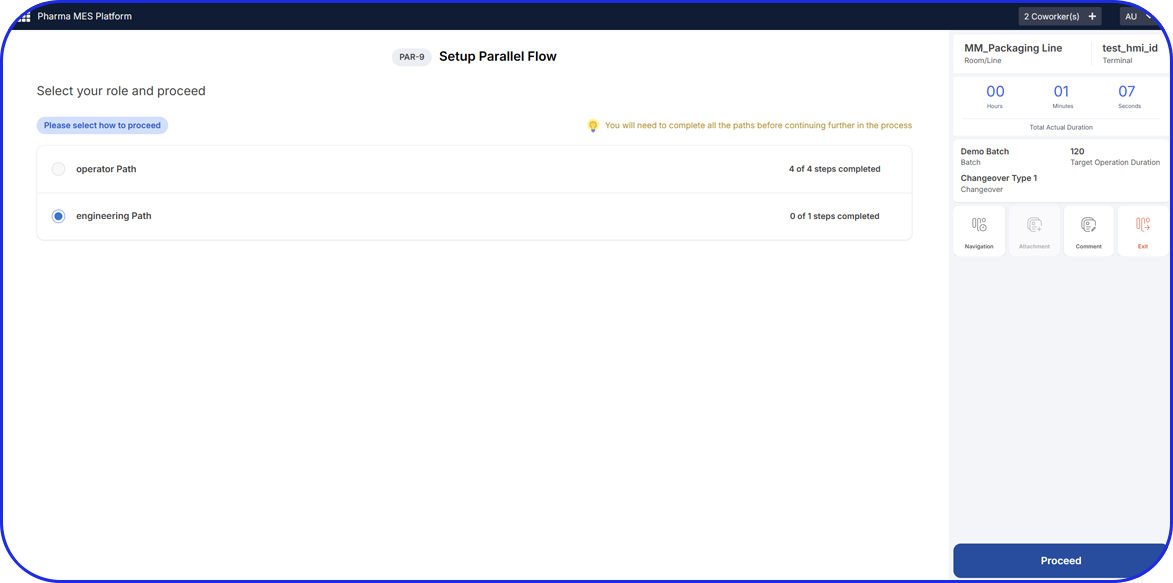
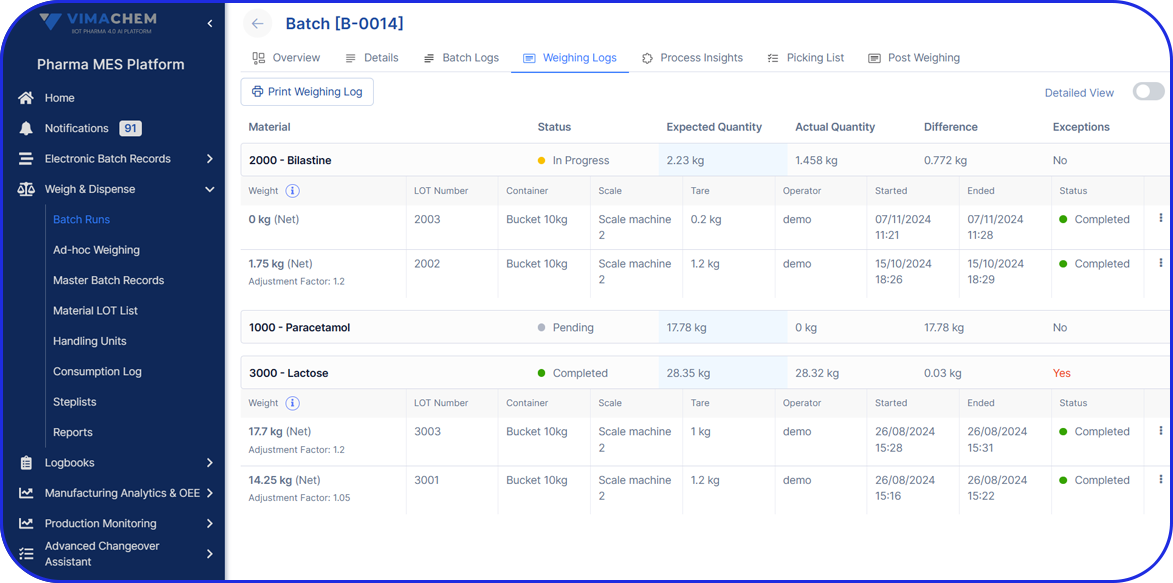
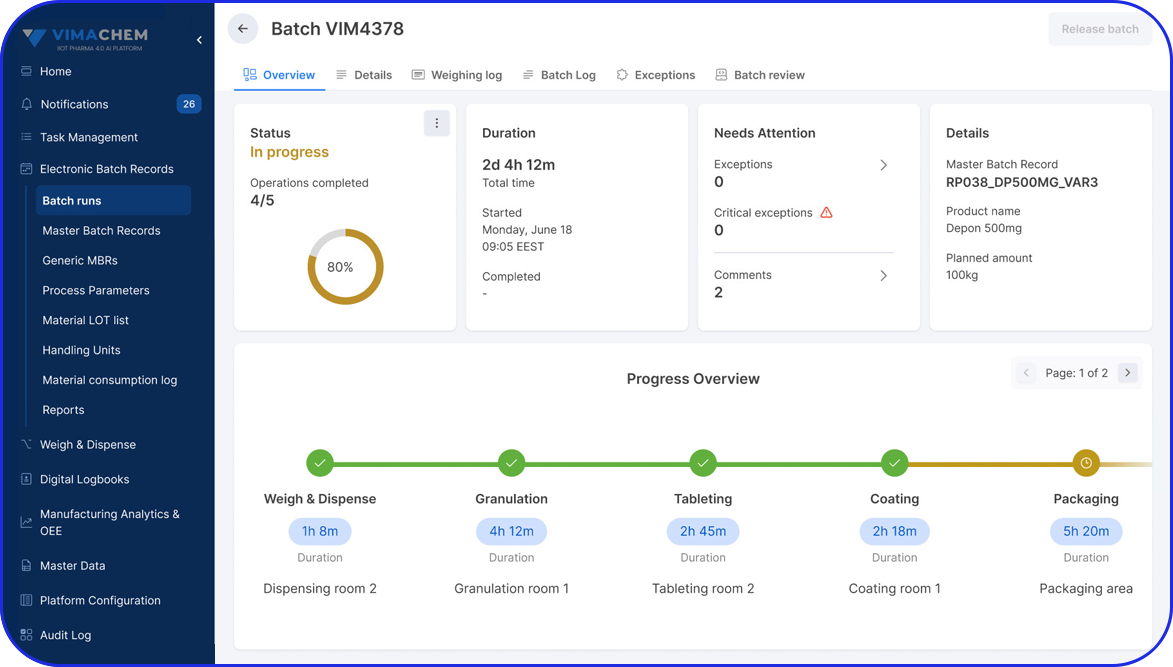
How Flexible is your Serialization Solution? How Flexible is your Serialization Solution? Vivi Korba Upstream Engineer https://youtu.be/PtLhYFWMPL8 Serialization is a constantly changing operation with new requirements (e.g., aggregation), evolving regulations (USA, UAE, etc.), and partner business updates, resulting in a constant increase in complexity, risk, and of course, costs. Multiple companies in the industry are […]
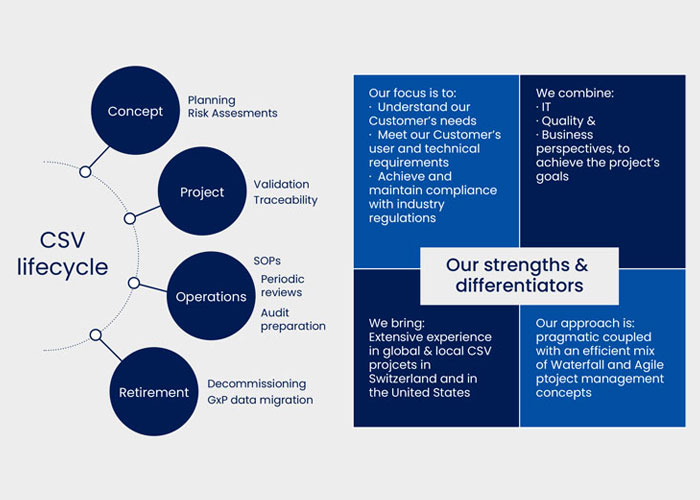
// Vimachem What Is Computer Software Assurance (CSA) – The FDA’s New Approach To CSV What Is Computer Software Assurance (CSA) – The FDA’s New Approach To CSV Anna Iliopoulou, MSc Quality Assurance and CSV Manager The purpose of pharmaceutical quality assurance is to ensure that the medication being manufactured will provide the desired effect […]
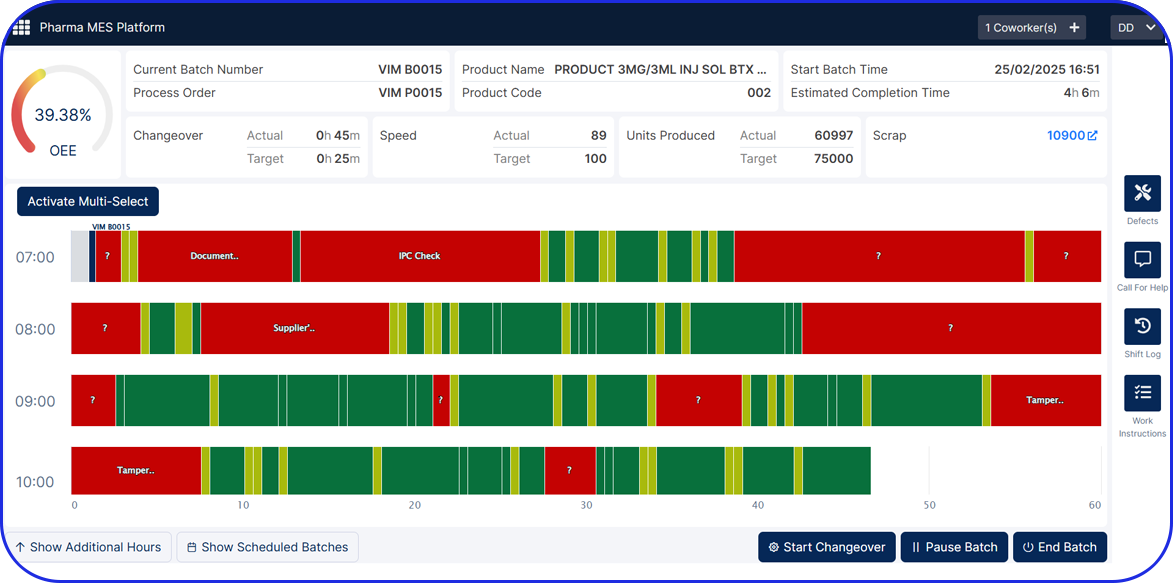
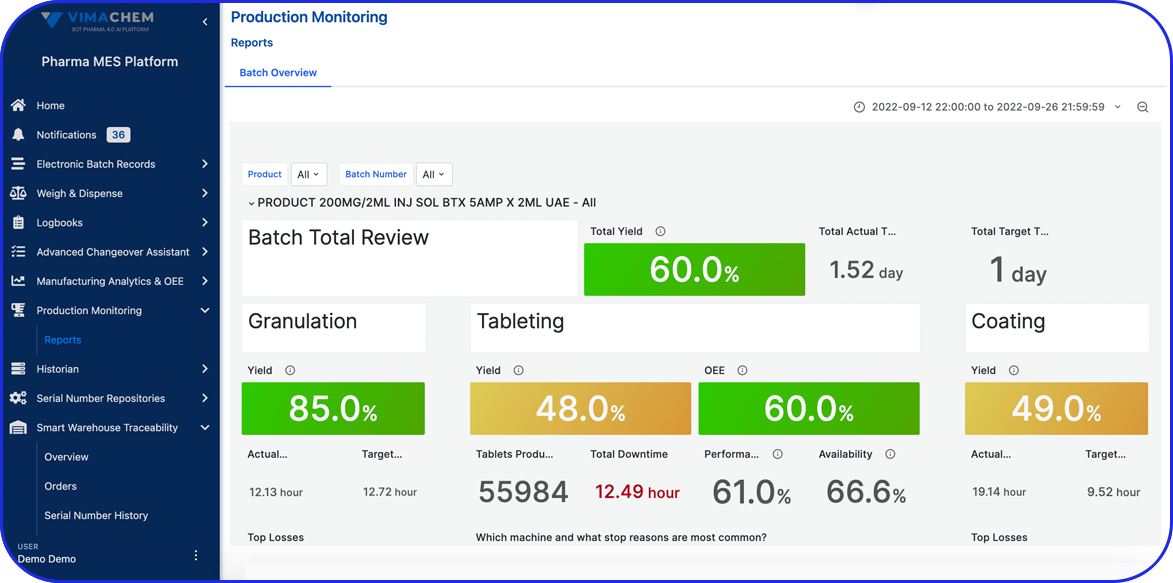
OEE As Your New ROI Standard In Pharma Manufacturing OEE As Your New ROI Standard In Pharma Manufacturing Olga-Maria Plessa Product Owner / Lead Consultant Return on Investment (ROI) has traditionally been the main driver for investments worldwide. Executives in pharmaceutical manufacturing have been utilizing ROI to evaluate and justify investment decisions. But how can […]
OEE Fundamentals OEE Fundamentals Olga-Maria Plessa Product Owner / Lead Consultant What is OEE and why is it important in pharmaceutical manufacturing? Fundamentals of OEE= availability, performance and quality Overall Equipment Effectiveness (OEE) Overview In an ideal pharmaceutical or biotech factory, equipment would operate 100% of the available time, at 100% capacity based on machines’ […]