Pharmaceutical Manufacturing Process: Key Steps Guide
Pharmaceutical Manufacturing Process: Key Steps Guide
The transformation of a pharmaceutical drug from a bold idea into a life-saving medication is an intricate and highly regulated process.
It’s a complex endeavor that requires ongoing collaboration of scientists, engineers, compliance, and quality assurance experts to ensure that all medications reaching the market are safe and effective.
Compared with less regulated industries, pharmaceutical manufacturing demands exceptional precision, control, and documentation.
Today, thanks to advanced digital manufacturing solutions such as AI-driven MES platforms with data-driven monitoring and analytics, drug production is becoming more efficient, adaptive, and aligned with Pharma 4.0 and digital transformation initiatives – ultimately ensuring safer, more reliable medications for patients.
What Is The Pharmaceutical Manufacturing Process?
The pharmaceutical manufacturing process is a series of meticulously controlled and regulated steps performed during the production of medicines. From the initial synthesis of active pharmaceutical ingredients (APIs) to the final dosage forms, e.g., tablets or injectables, it is designed to ensure that pharma products are safe, effective, and of consistent quality.
The Role Of Pharmaceutical Engineering
From research and development to manufacturing to post-marketing surveillance, pharmaceutical engineering plays a central role across the product lifecycle, from development through commercial manufacturing and lifecycle management.
Pharmaceutical engineering combines principles from chemical engineering, biomedical engineering, and pharmaceutical sciences to design, develop, and optimize processes and systems used in the pharmaceutical industry.
Pharma engineers design and establish manufacturing processes and production systems, meticulously designing every production step. They aim to streamline and optimize end-to-end drug manufacturing and, above all, ensure product safety and compliance.
The Pharmaceutical Process Design
Pharmaceutical process design, a key area of expertise, plays a critical role in drug production. Defined by pharma engineers, it focuses on the design of each step throughout the manufacturing process, pairing consistency and reproducibility with scalability and compliance with GMP (Good Manufacturing Practices) and other applicable regulatory requirements.
Pharma process designers continuously analyze data and adapt workflows to improve efficiency and reduce costs.
The Scale-Up Process In The Pharmaceutical Industry
One of the core aspects of pharmaceutical process design involves scaling up operations from the lab to commercialization.
Here, engineers evaluate various factors like equipment compatibility, reaction kinetics, and heat transfer to support a successful transition to commercial manufacturing while preserving product quality and process performance.
Pharmaceutical Manufacturing Is Generally Divided Into Two Main Stages
Following the R&D and clinical trial phases, the drug product is progressively scaled up for commercial manufacturing.
1. Primary Pharmaceutical Manufacturing
At this stage, the active pharmaceutical ingredient (API) is produced. Scientists and engineers focus on developing and manufacturing the API that provides the therapeutic effect. APIs can be produced at scale through chemical synthesis, fermentation, extraction from natural sources, or a combination of these.
APIs can be obtained in bulk quantities through chemical synthesis, fermentation, or extraction from natural sources – or a combination of these.
2. Secondary Pharmaceutical Manufacturing
During secondary manufacturing (drug product manufacturing), the API is formulated into a finished dosage form that patients can safely use.
Now, the API is combined with inactive ingredients called excipients to ensure its stability, bioavailability, taste, texture, shelf life, ease of consumption, and desired release profile within the body.
In a simplified scenario, once these two stages have been established, full-scale drug manufacturing can begin.
The Stages Of Pharmaceutical Manufacturing
It goes without saying that every type of drug has its own manufacturing methods. For example, a cough syrup wouldn’t require compression and coating like a tablet!
Nevertheless, pharmaceutical production follows some common steps, particularly for solid oral dosage forms:
1. Raw Material Procurement
The dedicated team evaluates qualified suppliers and applies the stringent quality criteria to select the ideal provider(s) of raw materials, be it synthetic chemicals or natural extracts.
Once materials reach the manufacturing plant, the quality control staff analyzes samples to ensure their purity and potency before they enter the production line.
2. Drug Product Manufacturing (Example: Solid Oral Dosage Forms)
Here, APIs and excipients are turned into the desired drug form.
Methods vary depending on the type of medicine, but they typically involve:
- Milling: Turning raw materials into fine particles for uniform mixing.
- Granulation: Forming powdered blends into granules for better flow and compressibility, either wet or dry.
- Mixing: Combining granules with excipients like binders, disintegrants, and lubricants.
- Tablet Pressing/Compression/Encapsulation: Compressing the mixture into tablets of desired shape and size, or filling them into capsules, ensuring uniform weight and dosage.
- Coating: Applying a protective layer to tablets for taste masking, controlled release, or moisture protection.
3. Quality Control & Testing
At every step of the manufacturing cycle, quality control personnel perform in-process control checks to verify that products meet the predefined standards for purity, potency, and uniformity.
This thorough testing ensures the safety, efficacy, and quality of every batch.
During and after packaging, additional quality checks such as weight verification, seal integrity, and visual inspection are performed to ensure consistency and compliance with specified requirements before dispatch.
While it’s impractical to test every single tablet or capsule, quality control units employ statistical sampling methods to effectively represent the entire batch.
4. Packaging, Labeling & Serialization
These are critical final stages in pharmaceutical manufacturing, ensuring the integrity, traceability, and proper usage of medications.
In a nutshell, the finished product is securely packaged in suitable containers that protect against contamination, light, moisture, and access by children, while also simplifying transport.
Then, every container is clearly labeled, following the legally required standards. Labels must include, among other things, information on dosage, indications, contraindications, potential side effects, and manufacturer details.
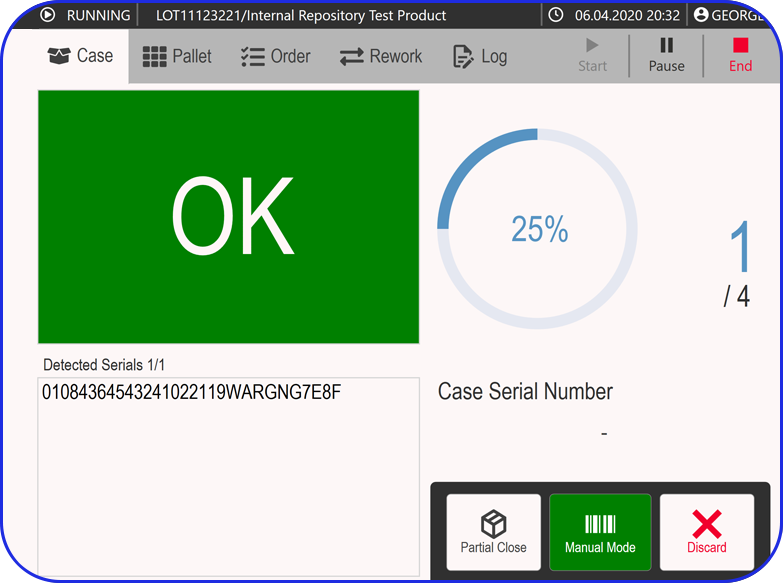
Finally, serialization is the process of assigning a unique serial number to each saleable unit of a pharmaceutical product, as well as its outer packaging.
Often in the form of a barcode, QR code, or another scannable format, pharmaceutical serialization provides a distinct identifier, so that every drug product can be tracked and traced throughout the entire supply chain, from production to the end-user.
It’s also a vital tool in the fight against counterfeit medicines, enabling verification of a product’s authenticity.
5. Distribution & Marketing
Once they leave the manufacturing plant, medicinal products are distributed through a network of wholesalers, pharmacies, hospitals, and physicians to reach patients in need.
This is a crucial stage for the commercial success of the drug and follows different marketing strategies and educational campaigns to inform both healthcare providers and potential users about the new medication and its benefits.
On top of mere sales, this stage aims to help different types of medicines reach the patients who need them. It also requires defined transport and storage conditions to preserve drug quality and effectiveness throughout the supply chain.
6. Post-Marketing Surveillance
Even after a drug hits the market, it’s continuously monitored for any long-term side effects or adverse reactions that may not have been detected in clinical trials. Post-market surveillance and risk management plans are vital and also mandatory.
Using real-world evidence (RWE) from sources such as electronic health records, often aided by advanced data analytics and artificial intelligence, researchers essentially identify and address any issues that arise as diverse patient populations use drugs over extended periods in real-world settings.
At this stage, pharmaceutical companies are required to monitor and report any unanticipated experiences or side effects associated with their products – a process known as adverse event reporting. In such cases, drug product serialization supports rapid traceability and targeted recalls when safety issues emerge.
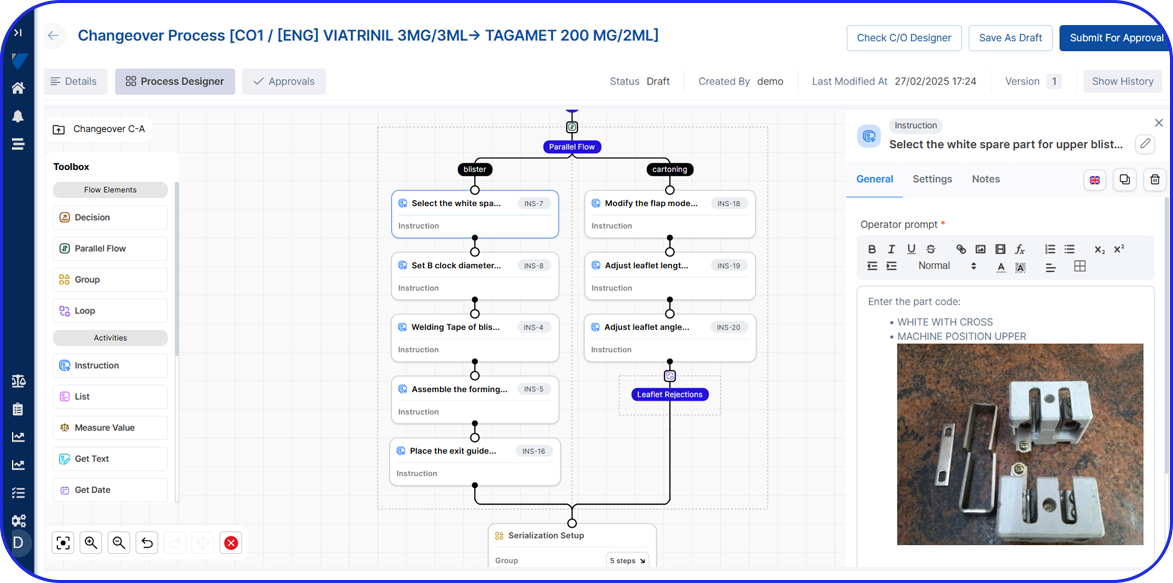
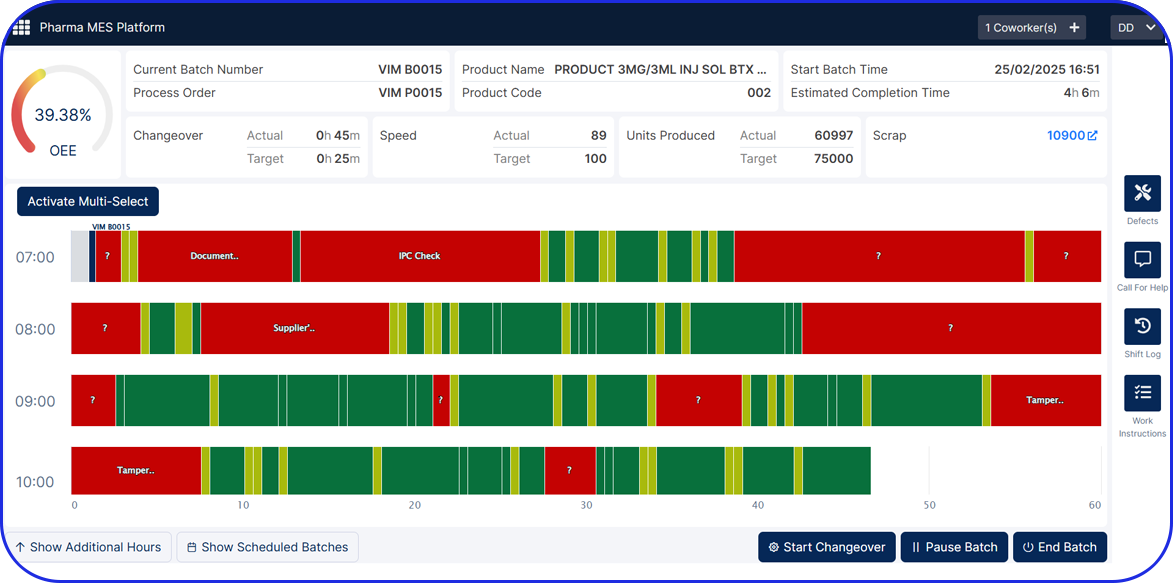
Streamlining Pharmaceutical Manufacturing For Pharma 4.0
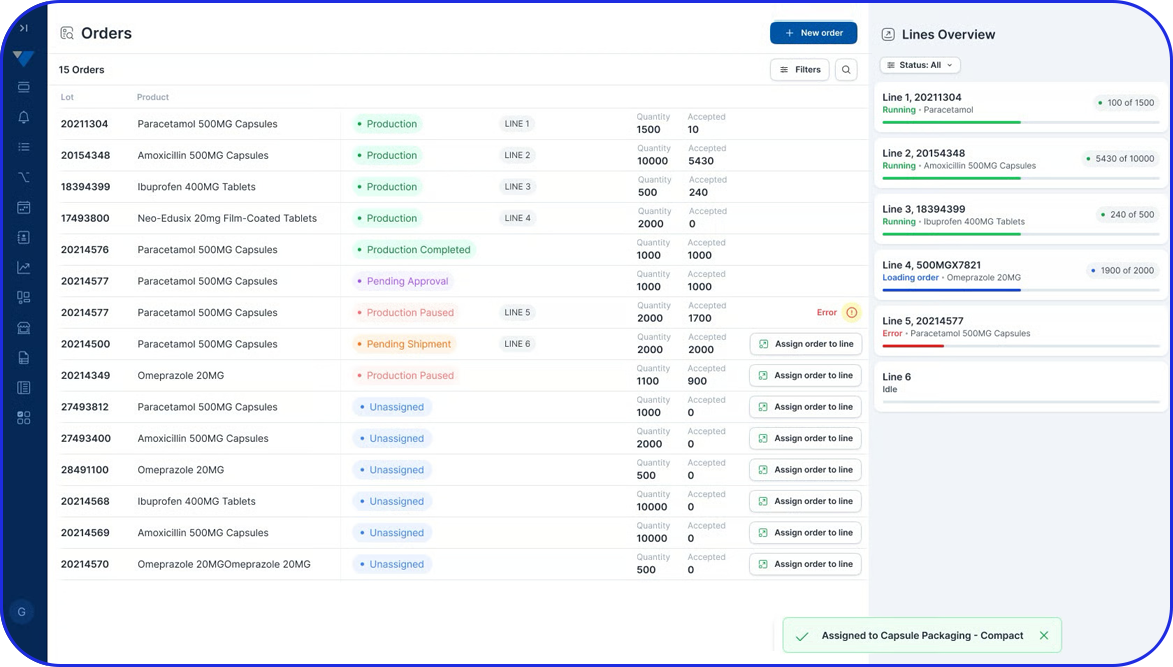
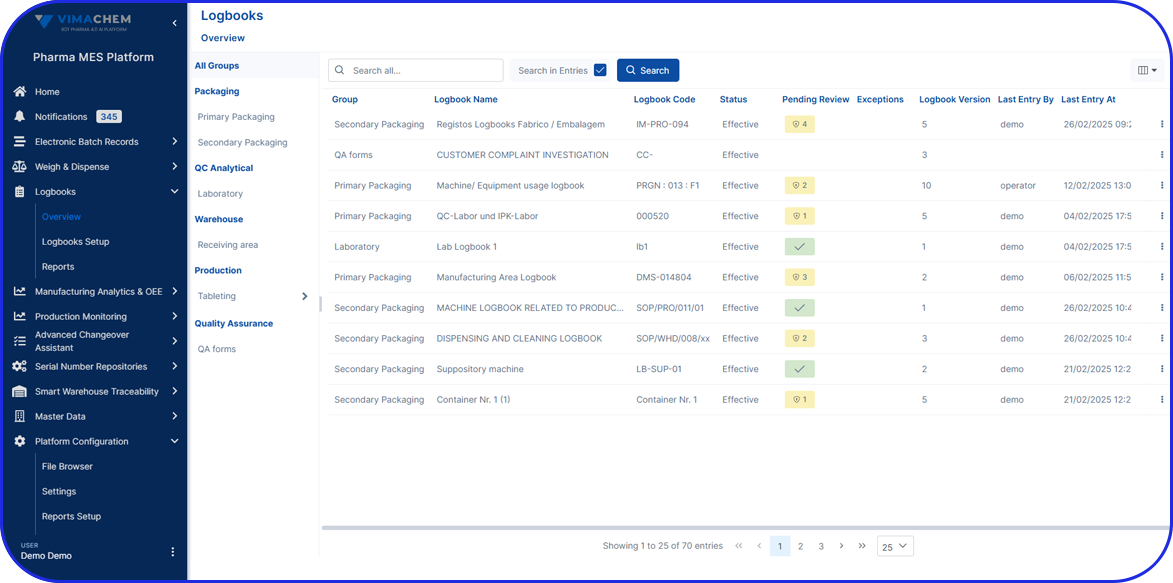
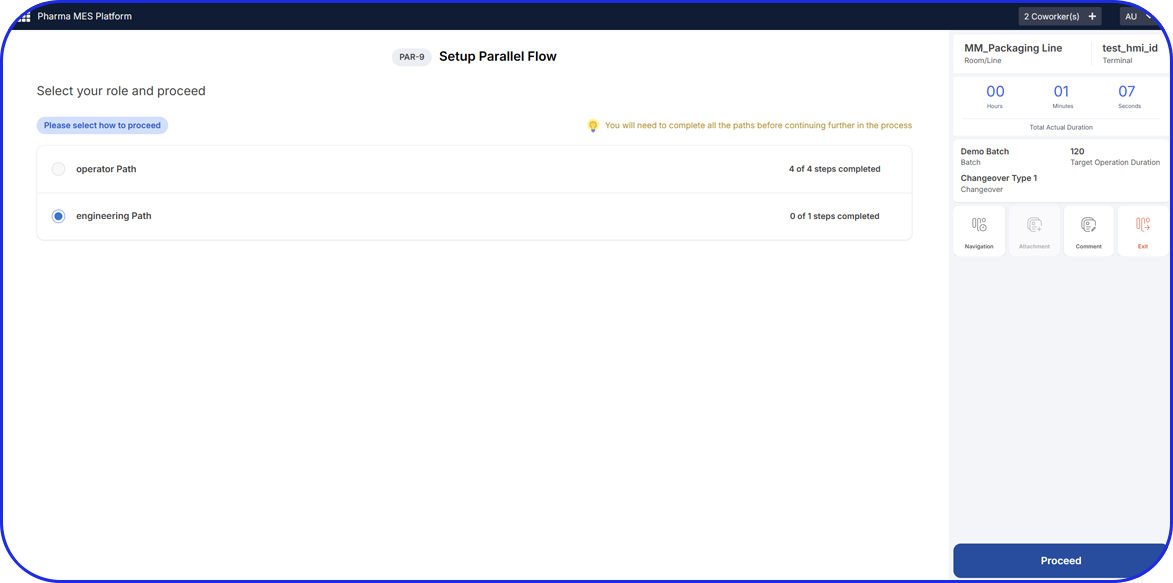
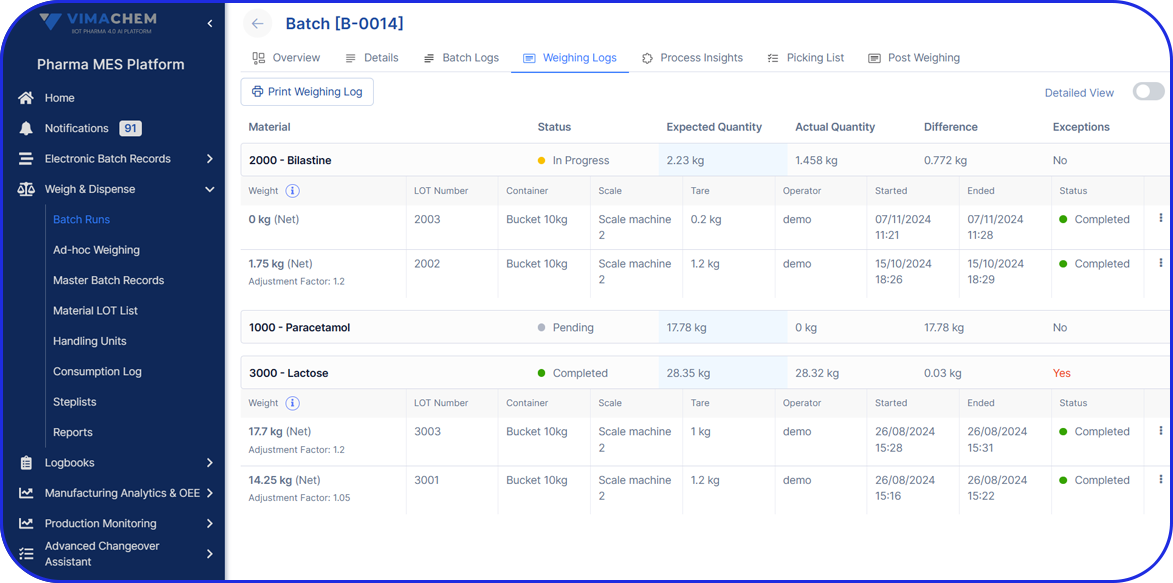
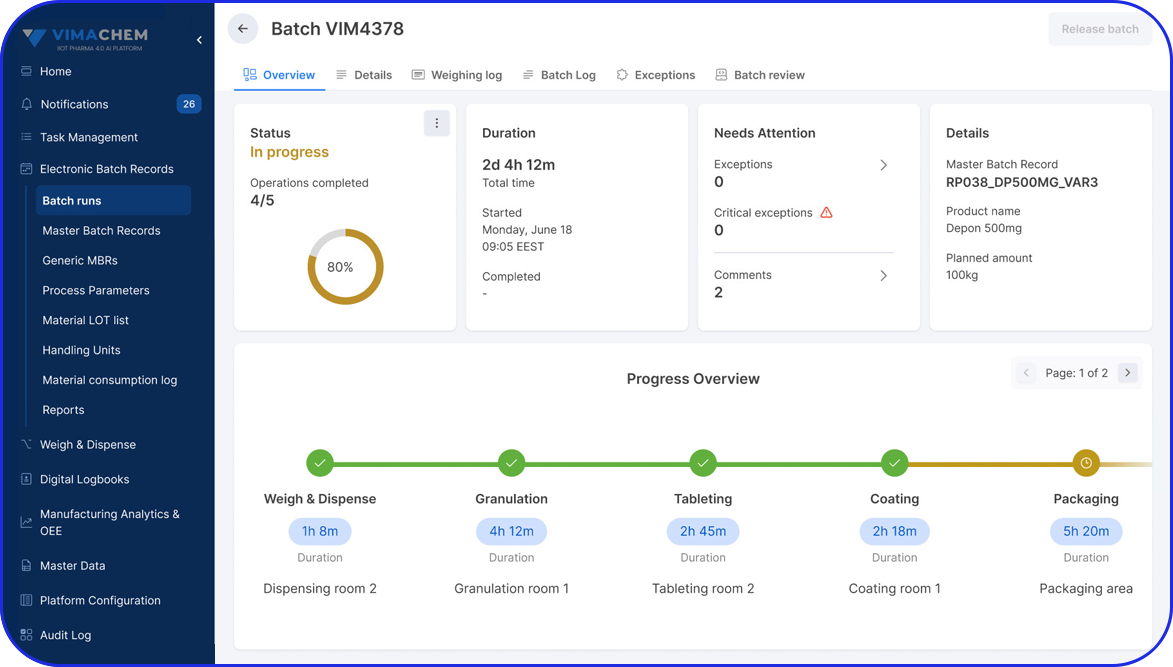
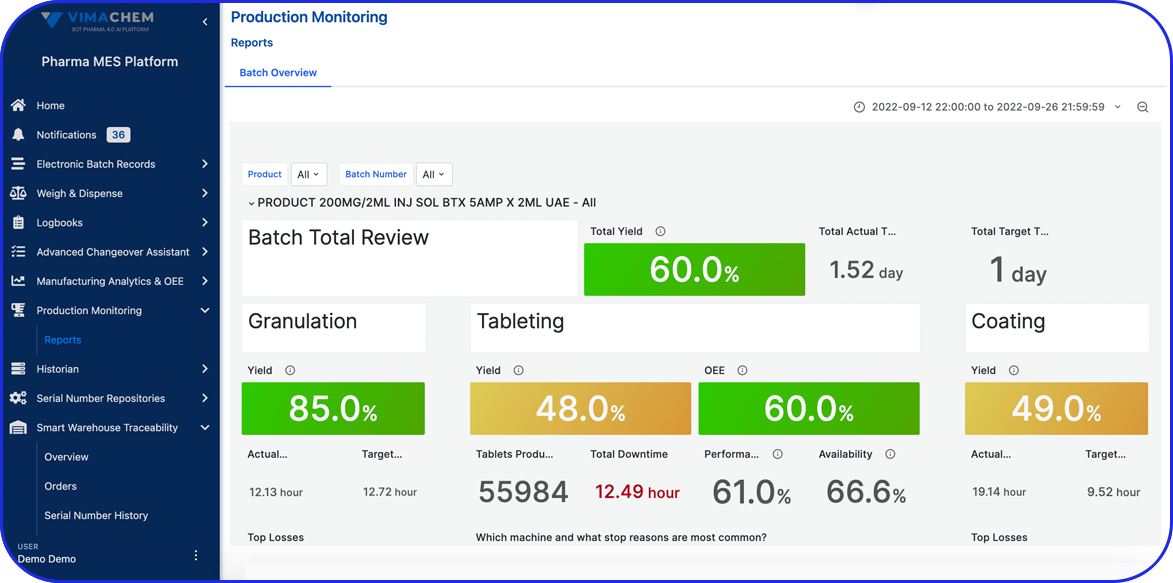
As pharma production transitions into Pharma 4.0, Vimachem’s AI-driven MES Platform stands at the forefront of the digital transformation.
Designed specifically for pharma and biopharma, Vimachem’s MES platform integrates advanced analytics, AI-enabled capabilities, and IIoT connectivity into manufacturing operations.
Driving the digitalization journey, Vimachem MES provides real-time contextualized feedback, enabling seamless progression from planning to product delivery, while steadfastly maintaining cGMP and 21 CFR Part 11 compliance.