A Guide To Pharmaceutical Serialization: All You Need To Know
A Guide To Pharmaceutical Serialization: All You Need To Know
The pharmaceutical industry operates within complex supply chains. From production to the end consumer, prescription drugs pass through various entities, increasing the risk of counterfeit medicines infiltrating the market.
These fraudulent drugs may be contaminated, contain incorrect or inactive ingredients, or wrong dosages. They pose serious health dangers and can be fatal.
Moreover, pharmaceutical companies lose billions annually due to grey market medicines and counterfeits.
To combat these threats, pharmaceutical serialization and traceability play a crucial role in ensuring drug authenticity and protecting public health. Advanced pharma serialization software helps manufacturers streamline compliance, enhance tracking, and strengthen supply chain security.
What is Pharmaceutical Serialization?
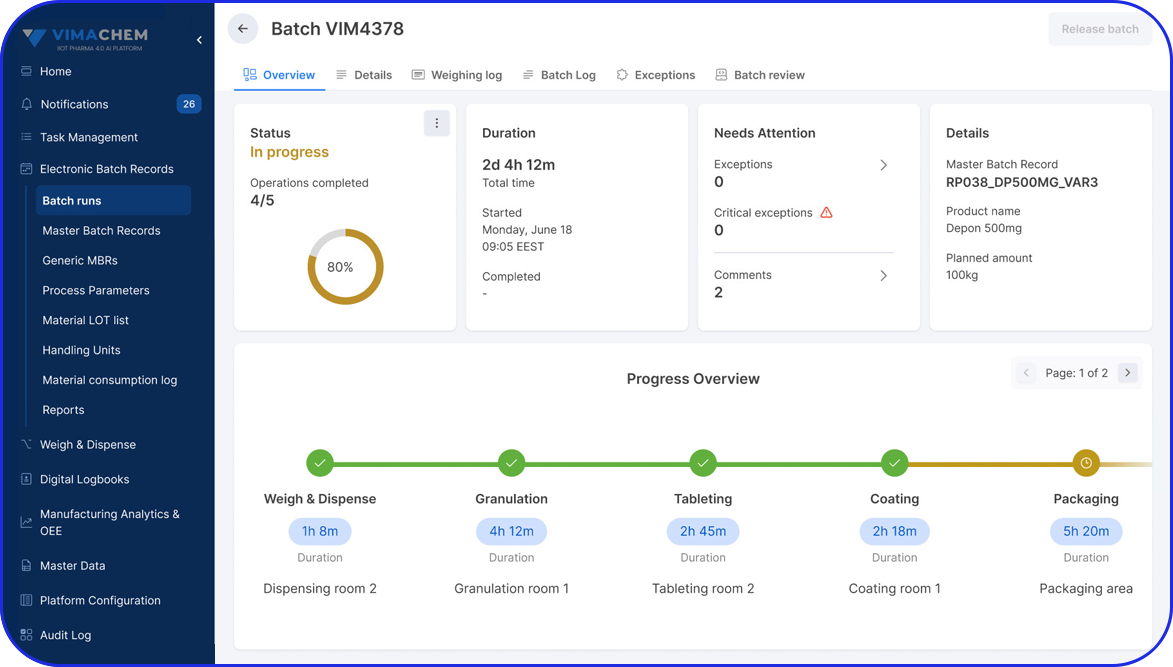
Pharma serialization involves creating a unique identifier for each pharmaceutical product and printing this code on the label or packaging before distribution.
The unique identification code enables end-to-end tracking of every unit produced throughout the supply chain, from the manufacturing site to the pharmacy or medical center.
Today, serialization in pharmaceutical packaging is imperative due to growing concerns about drug counterfeiting, diversion, and theft.
To facilitate track and trace, serialization labels of every saleable unit typically include a unique serial number, lot number, identification code, batch number, and expiration date. However, different countries may have different requirements and registration policies.
How does Serialization work?
Before a drug reaches the market, manufacturers serialize each unit by printing a unique identifier,such as a barcode, QR code, or another scannable format, on every package. This serial number, along with other relevant data, is then recorded in a central database.
Serialization enables product tracking and verification at every stage of the supply chain, facilitating the removal of counterfeit or stolen drugs before they reach patients.
It also ensures that pharmaceutical drugs are stored and transported under proper temperature and environmental conditions, safeguarding patient safety.
A more granular level of serialization enables traceability down to individual tablets or capsules through unique codes, which can be linked to the primary packaging and even the pallet.
Last year in the US, the FDA passed a new Drug Supply Chain Security Act (DSCSA), that enhanced serialization requirements for pharmaceuticals, taking advantage of automated data management and cutting-edge cloud storage technology. The goal was to ensure secure product traceability, enable package-level verification, and allow for a swift response to illegitimate or suspect products.
But other countries are also upgrading their requirements for the serialization of pharmaceutical products – each with its own nuances. As a result, Pharma manufacturers operating internationally must navigate complex regulatory landscapes.
New US Serialization Requirements: the road to DSCSA Compliance
The DSCSA introduces stringent serialization requirements to secure the pharmaceutical supply chain. Title II emphasizes the traceability of prescription drugs at the package level, ensuring the identification and removal of illegitimate products. Key technical aspects include:
- Interoperable Data Exchange: Manufacturers, distributors, and dispensers must integrate systems for seamless electronic data sharing.
- Product Verification Systems: Automated verification systems to check product identifiers against serialized databases.
- Aggregation Requirements: Enhanced aggregation of serial numbers for pallets, cases, and individual units to streamline tracking and recalls.
Changes to the EU Falsified Medicines Directive (FMD): Implications for Serialization
The upcoming FMD changes introduce advanced technical demands for compliance. Key changes include:
- Mandatory Digital Reporting: Real-time data uploads to national repositories for authentication and traceability.
- Enhanced Serialization Elements: Integration of GS1 Digital Link standards for expanded data storage within barcodes, including dosage and administration instructions.
- Tamper-Evident Features: Mandatory implementation of secure packaging solutions to prevent unauthorized access or modifications.
- Integration with Blockchain: Some EU states are exploring blockchain to create immutable records for tracking pharmaceuticals across borders.
How Pharmaceutical Serialization Helps
Serialization in Pharma supports public health and the pharma market in various ways:
Combats Deaths Caused by Counterfeit Drugs
The global growth of counterfeit drug trading is alarming, estimated at a $4.4 value worldwide in 2023. This illegal but high-profit margin activity causes more than 1 million deaths per year, according to the World Health Organization.
With families mourning loved ones lost to counterfeit cough syrup or pain relievers, strengthening and enforcing medication serialization is a critical public health priority.
Protects Lost Jobs, Revenue and Innovation
The production of counterfeit drugs is a worldwide problem, but top manufacturing countries suffer from huge drops in revenue. In the US alone, the pharmaceutical sector is losing up to $162.1 billion in revenue every year, plus billions in federal tax.
This unavoidably translates into tens of thousands of lost jobs and reduced pharmaceutical innovation.
It is estimated that for every $100 billion global counterfeit medicine sales, there’s a $17 billion loss in research and development projects, 6 less new drugs, and more than 57,000 lost jobs.
Enhances Transparency and Traceability
Technological advancements enable more sophisticated and intelligent pharma serialization procedures, ensuring end-to-end transparency of prescribed drugs throughout the entire manufacturing and distribution journey.
This allows for a detailed investigation to pinpoint where a suspect product entered the supply chain, further safeguarding the market from dangerous pharmaceuticals.
Verifies Trading Partners
Current serialization practices offer an extra layer of protection for hospitals, clinics, and pharmacies. Using specialized connected platforms, they are able (or often required) to verify that their suppliers are authorized trading partners in good standing, safeguarding their patients’ health and also their brand name.
Co-Ordinating Product Recalls
From time to time, certain pharmaceutical products or a series of drug batches may present unforeseen concerns.
In such cases, serialization databases significantly streamline mass recalls, enabling fast and effective tracking of problematic units—from the production plant to pharmacy shelves and hospital cabinets worldwide.
Making Strategic Manufacturing Decisions
An effective drug serialization strategy provides pharmaceutical manufacturers with detailed insights into their supply chain and consumer behavior, enabling them to make informed decisions on their procurement, production and innovation strategies.
Despite all the benefits of serialization, new, intricate compliance requirements present diverse challenges for pharmaceutical manufacturers.
Smarter serialization, seamless compliance.
Navigating Serialization Challenges with Vimachem
Pharma serialization regulations are constantly evolving, requiring pharmaceutical manufacturers to adopt robust and adaptable platforms to ensure compliance both nationally and internationally.
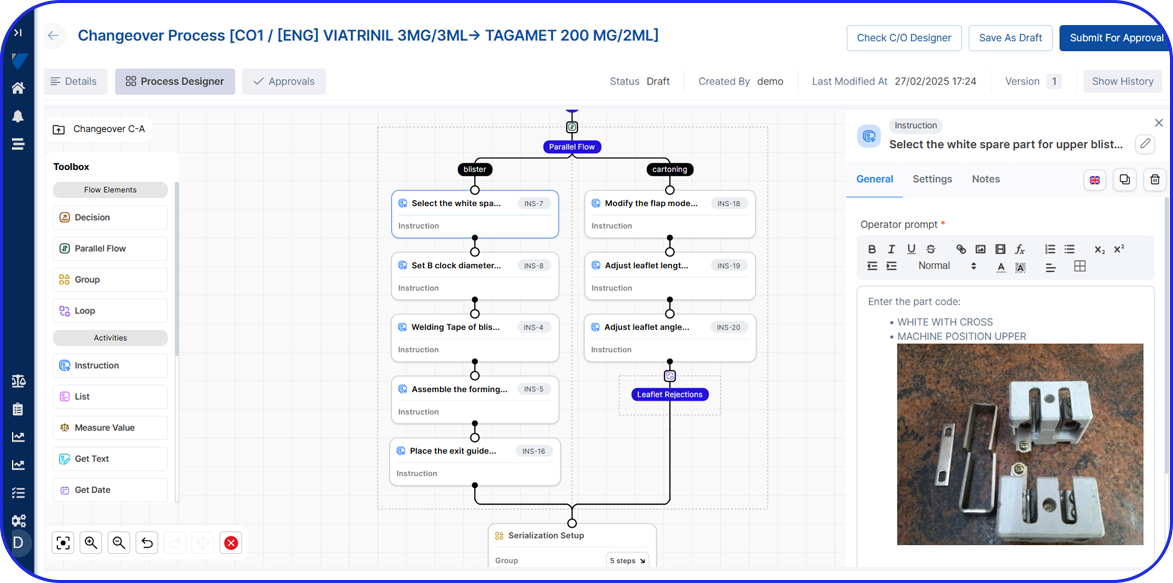
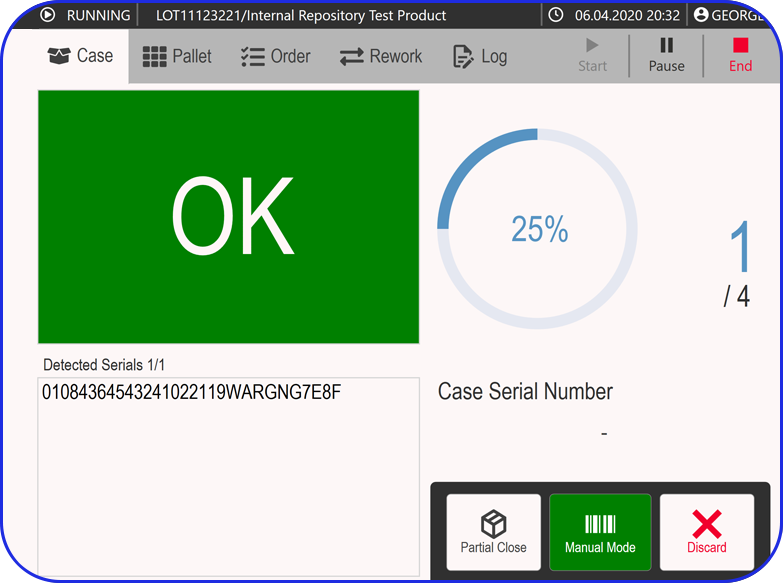
Vimachem Level 3 Serialization Software is a cutting-edge traceability solution that seamlessly integrates with any production line or network system, ensuring compliance with global serialization requirements.
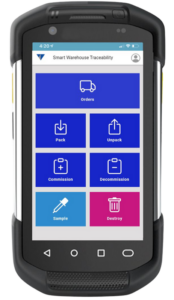
Designed as a best-in-class serialization software, it overcomes common challenges such as vendor lock-in and slow response times, eliminating incorrectly coded or packaged products while enhancing production output and packaging OEE. It also seamlessly integrates with Vimachem’s Smart Warehouse Traceability (SWT) solution, which supports Third-Party Logistics (3PLs), wholesalers and parallel distributors, providing customers with end-to-end visibility, while ensuring compliance, and enhancing efficiency.
Vimachem’s solution stands as the world’s largest line and network-agnostic track-and-trace software in pharmaceutical manufacturing.
Contact us
Ready To Augment Your Shop Floor Operations?
Ready To Augment Your Shop Floor Operations?
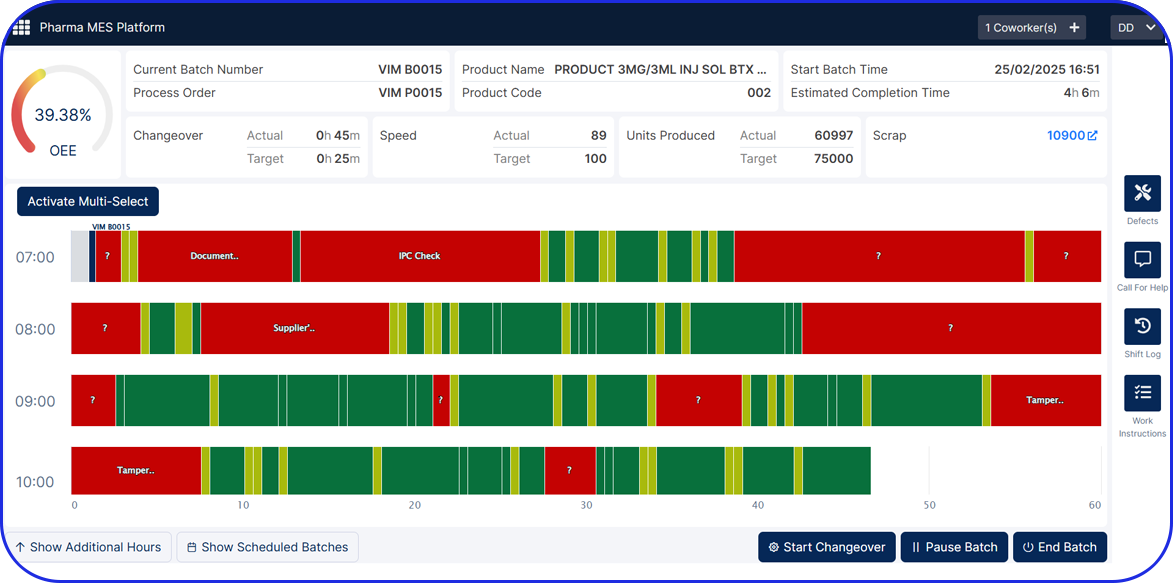
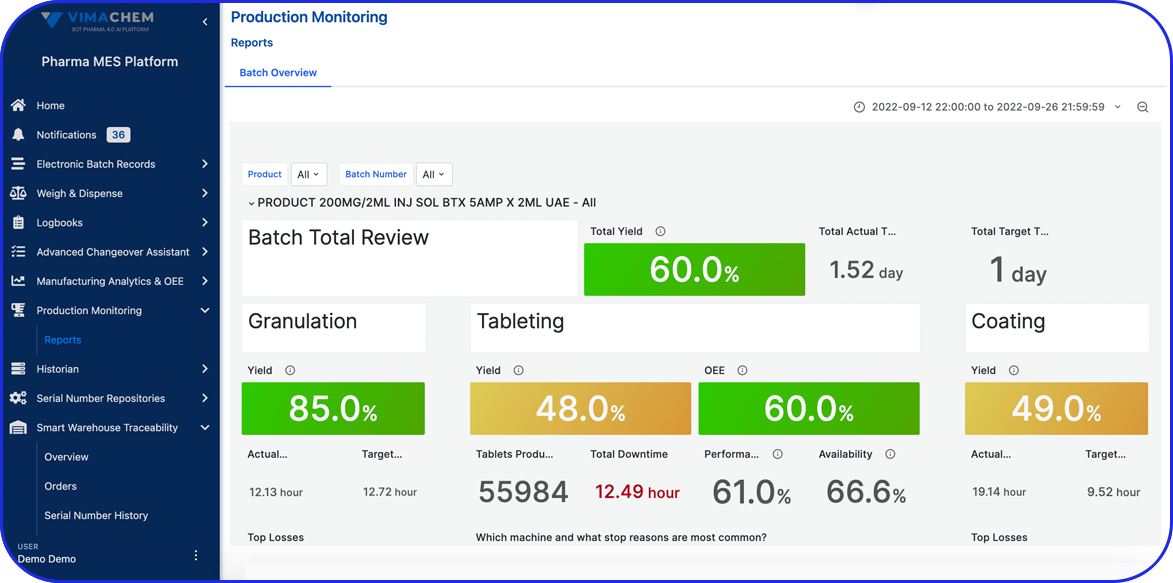
Vimachem Manufacturing Analytics - OEE
The Manufacturing Analytics – OEE module is an intelligent Pharma OEE Cloud solution that allows you to collect, store and visualize data across your site / enterprise and apply AI algorithms to optimize production efficiency and product quality.