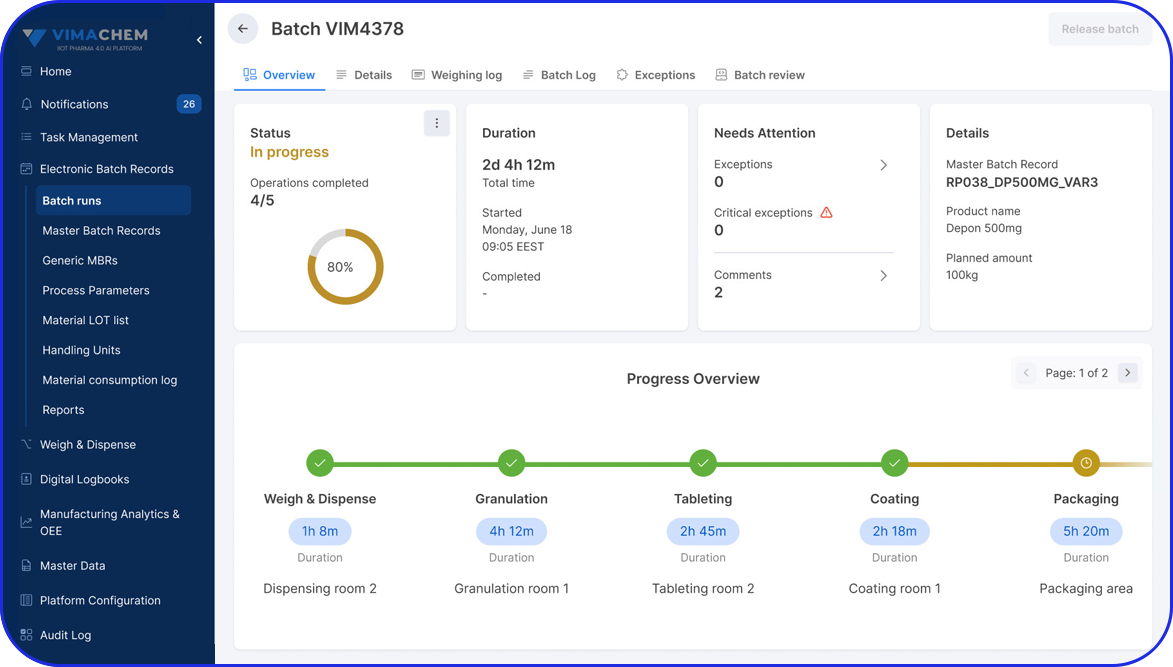
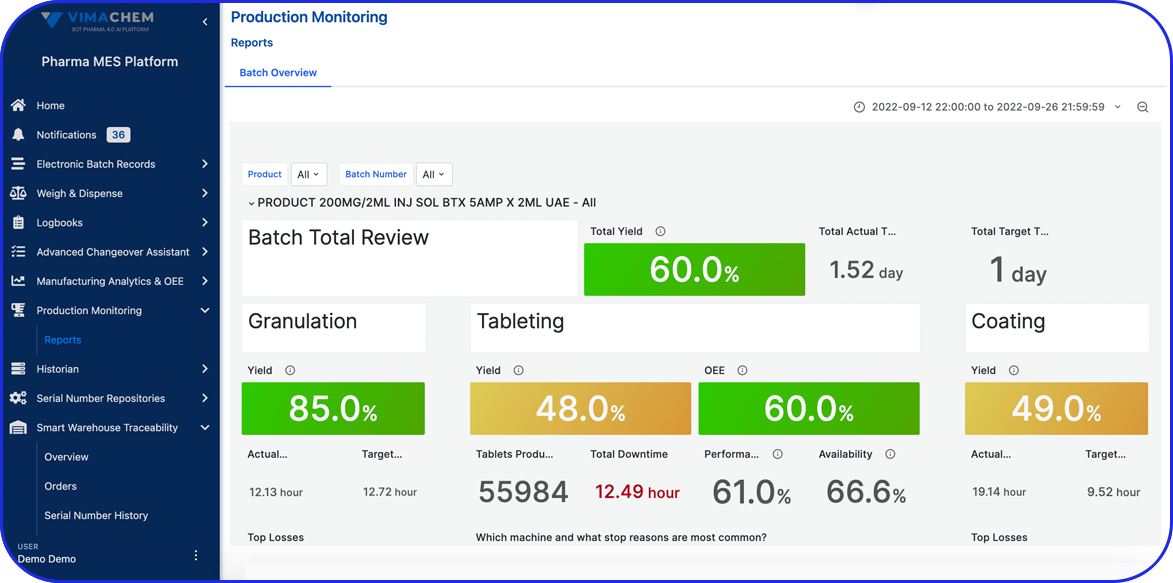
Batch vs Continuous Process in Pharma Manufacturing
Batch vs Continuous Process in Pharma Manufacturing
However, as the digitalization of pharma and biopharma leverages innovative technologies like the Internet of Things, artificial intelligence, and big data, the shift towards continuous process manufacturing is gaining momentum.
Let’s take a closer look.
What Is Meant By Batch Process?
A batch process is a rather traditional manufacturing method where goods are produced in defined quantities, following distinct, sequential steps. Each batch progresses through defined stages in the production cycle before moving to the next step. This staged approach involves pauses for activities such as in-process testing, material handling, storage, and equipment preparation between steps.
Batch processing is straightforward. It is backed by extensive knowledge and technology, and, in regulated industries like pharma, it needs to follow an established set of compliance guidelines.
Characteristics Of Batch Manufacturing
Here are some key features of batch production:
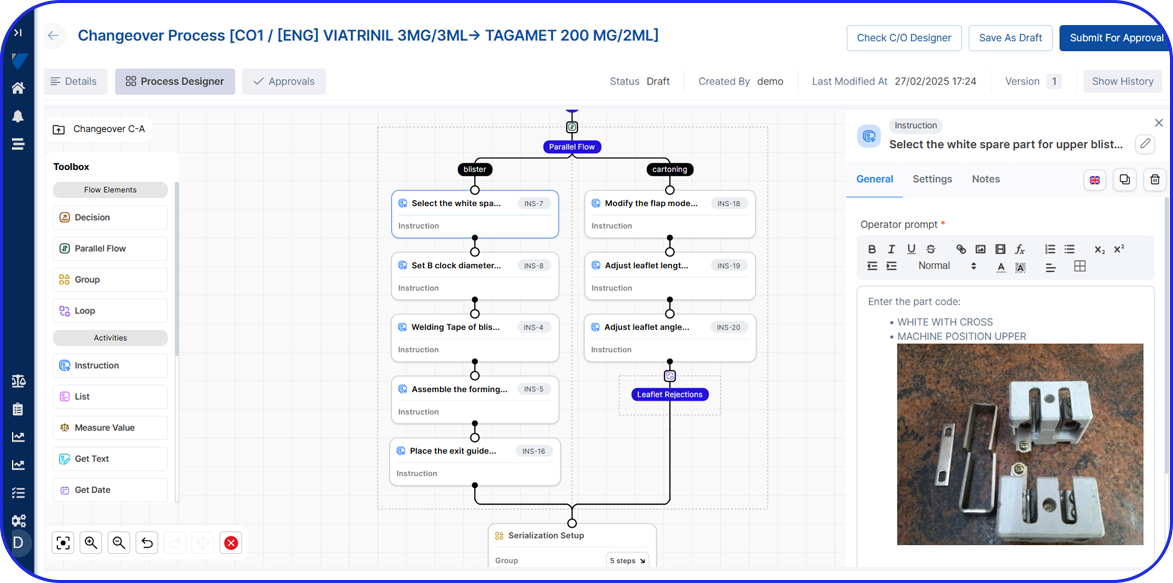
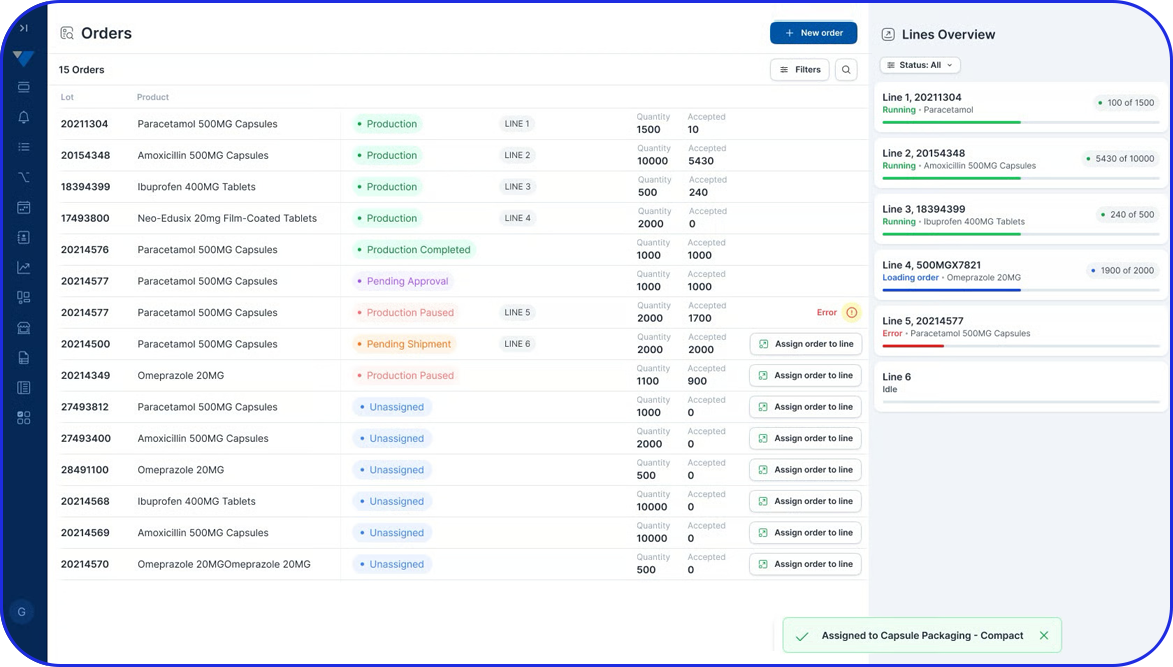
- Sequential Processing: Production follows a standard procedure divided into concrete stages. For example, raw materials for a particular batch typically move to the next stage after processing in the current stage is complete.
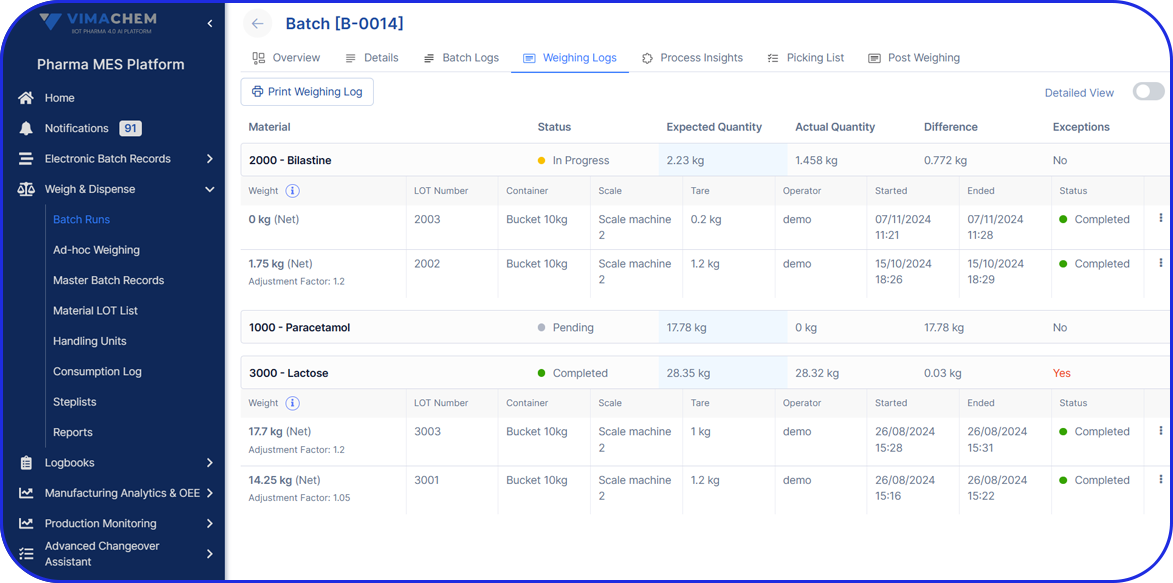
- Quality Control Points: Quality checks in batch manufacturing include in-process controls during production and final testing at the end of each batch. So, if there are any issues, there will be the appropriate quality adjustments made to the subsequent batches to maintain high standards.
- Flexibility & Customization: Due to its granular setup, batch processing allows manufacturers to reconfigure equipment to produce diverse product lines or customized formulations.
- Resource & Time Consumption: Batch manufacturing typically consumes significant time and resources as it gradually moves from raw material to finished products. It often requires handling, storing, and monitoring intermediate products between phases, thus extending production time and costs, and, in some cases, the risk of variability.
What Is Continuous Process In Pharma Production?
Continuous process manufacturing in pharma is a fairly new production method that follows an uninterrupted flow. Here, raw materials are continuously fed into one end of the system, and finished product is produced continuously during extended manufacturing runs.
Continuous manufacturing (CM) has long been used in other industries, especially those with low customization needs. Advances in process control, PAT (process analytical technology), integrated equipment design, and regulatory frameworks have enabled the adoption of continuous manufacturing in pharmaceutical and biopharmaceutical production.
Such flow production operations can enhance efficiency on multiple levels, as they typically lead to reduced processing time, energy consumption, and running costs. Also, in many cases, they can help combat market shortages of vital medicinal drugs.
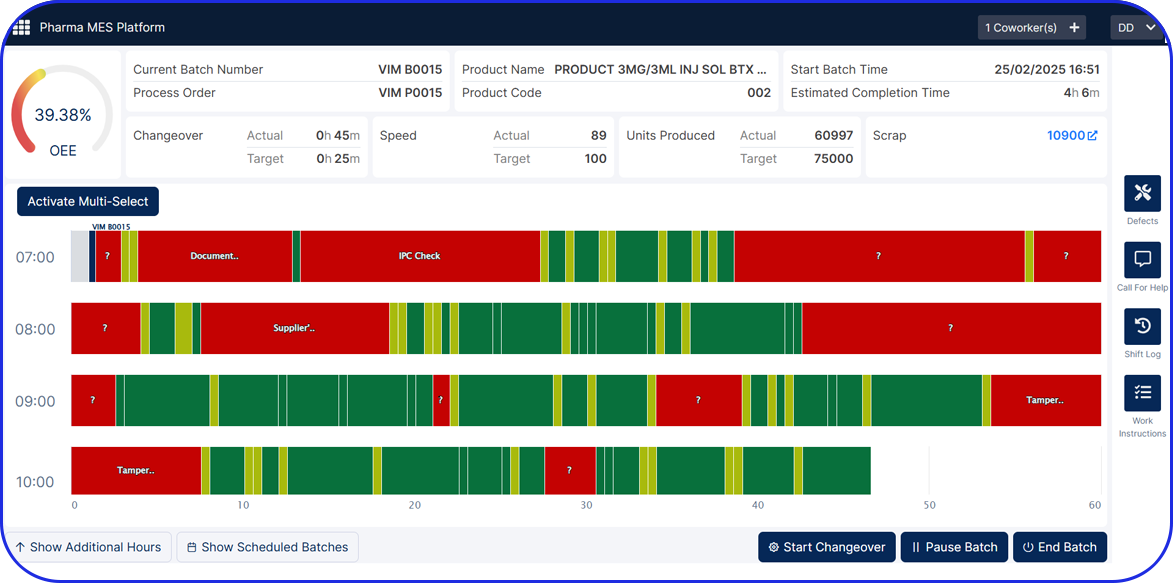
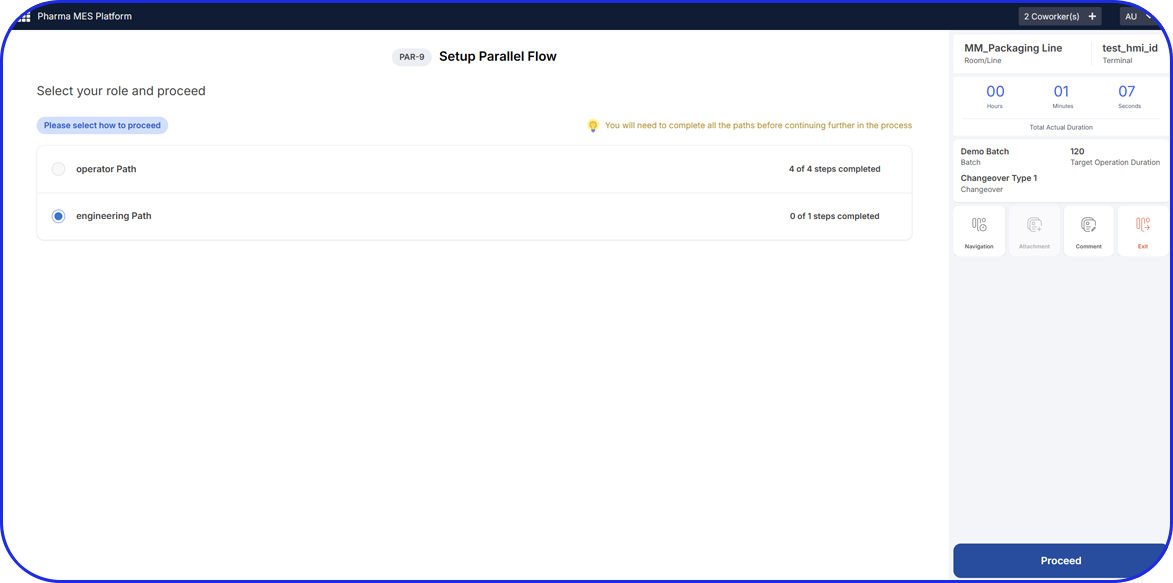
In pharma production, continuous process systems are designed explicitly for high consistency and typically operate with minimal human involvement, relying heavily on automated systems, like AI-Driven MES (Manufacturing Execution Systems).
Batch vs Continuous Process - Key Differences
The core difference between batch and continuous processes lies in whether products are made in self-contained phases or a continuous, uninterrupted flow. This differentiation carries several important implications for pharmaceutical manufacturing.
Production Rate
Producing in batches, unavoidably, has lower throughput, as operations halt between runs. In contrast, continuous manufacturing has shorter processing times that may in some cases significantly reduce manufacturing cycle times, potentially shortening production timelines from weeks to days depending on the product and process design.
Flexibility
Thanks to their interruptibility, batch processes are highly flexible, allowing for easy adaptation to varying customer demands and product changes between runs. But the continuous model offers limited flexibility. Customizations and changes often require substantial investment in new equipment. That’s why it’s currently more suitable for specific drug product types, like certain tablets or capsules.
Quality Control
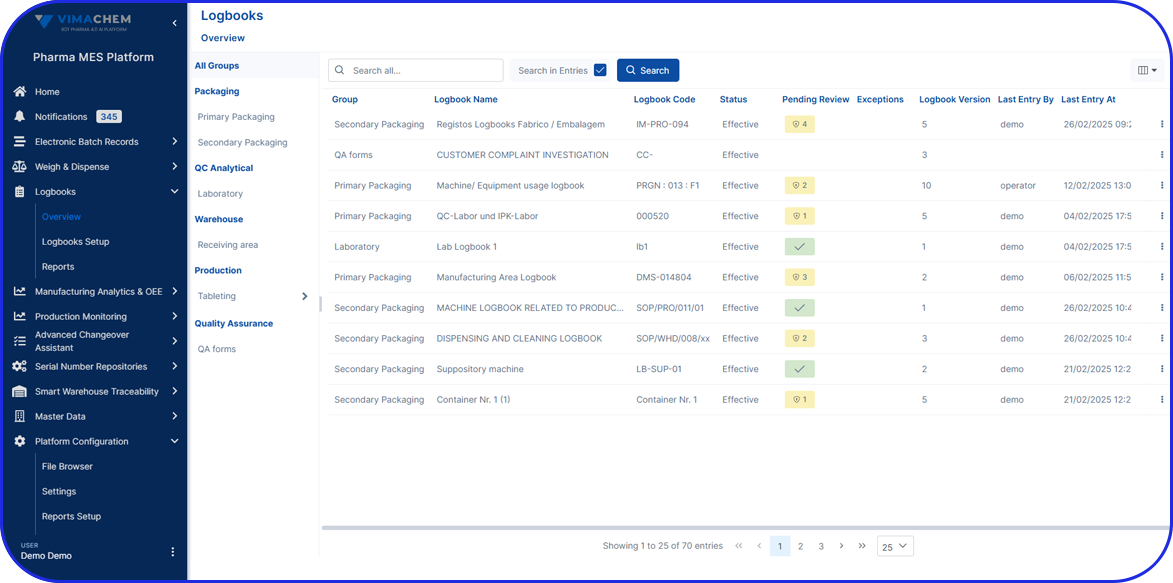
Traditionally, quality checks in batch production were conducted at the end of each batch. However, thanks to today’s technology, organizations can integrate EBR (Electronic Batch Records) within an AI-driven Manufacturing Execution System (MES) and conduct quality checks throughout every production stage of a certain batch.
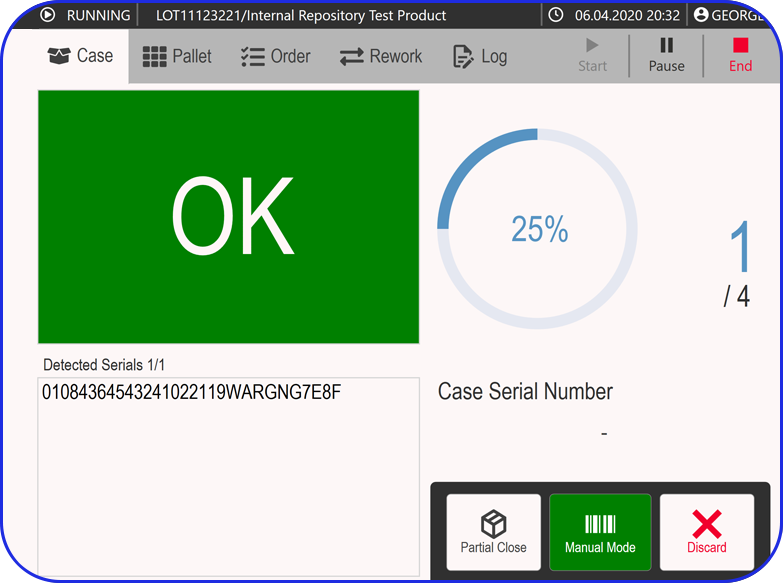
Continuous processes take QC a step further. With the help of intelligent, automated systems for end-to-end real-time monitoring, there can be immediate detection and correction of any deviations. For example, advanced monitoring with process analytical technology allows for increased sampling frequency without requiring physical sample collection.
Equipment & Maintenance
Batch processing equipment is generally simpler, smaller, and easier to maintain on a periodic basis. On the other hand, CM systems are more sophisticated, complex, and costly, requiring robust and frequent maintenance protocols due to continuous wear and tear.
Cost Considerations
While batch processing has lower initial setup costs, it typically incurs higher operational expenses due to frequent setups, cleaning, and interruptions.
CM demands a significant initial investment for specialized machinery, which is often offset by lower unit costs achieved through higher production rates, reduced waste, and energy consumption over the long term.
Research indicates that continuous manufacturing can reduce product variations by 50%, the time required for quality control by approx. 60% and power consumption by 40%.
Human Error & Contamination
Continuous process’s high level of automation significantly reduces manual handling and the risk of human error, leading to improved consistency. The closed nature of these systems also minimizes the risk of product leakage or contamination during transfers, which are common in multi-site batch operations.
Supply Chain Management
Batch production can have sourcing, handling, and even production stages dispersed across different sites and locations. However, continuous processing often integrates multiple drug-product steps within a single facility, though API manufacturing and packaging may still occur separately depending on the product and supply chain design.
Continuous Or Batch Production? Future Trends In Pharma Manufacturing
Different analysts and industry experts support the expansion of continuous manufacturing in pharma production.
According to a research effort led by Clifford Rossi at the University of Maryland, “CM appears to win handily over batch processing in the U.S. and even provides an economic advantage over batch processing abroad”.
Similarly, Ron Piervincenzi, Ph.D., CEO of USP, emphasizes that “Within the next decade, PCM is poised to become an industry staple alongside traditional batch manufacturing for the production of both innovative and generic pharmaceuticals and biologic products”.
Despite these clear advantages, continuous manufacturing is adopted at a rather slow pace. This is most probably due to the significant initial investment costs, integration complexities, and the need for specialized expertise.
However, the demand for supply chain resilience -intensified by adverse events like the COVID-19 pandemic is accelerating interest in continuous manufacturing in an effort to strengthen domestic production of essential medicines.
As for the future of pharma manufacturing, it’s believed that a hybrid system, combining elements of both batch and continuous processes, will prevail in the coming years.
How Vimachem’s AI-Driven Solutions Can Help
Navigating the complexities of modern pharmaceutical manufacturing, whether batch, continuous, or semi-batch, demands advanced technological solutions.
Vimachem’s IIoT AI-Driven MES platform offers a comprehensive suite of tools designed to optimize production, ensure compliance, and unlock the full value of manufacturing data, helping companies enhance process optimization and achieve faster time-to-market.