What Is A Batch Record? Role & Examples In The Pharma Industry
What Is A Batch Record? Role & Examples In The Pharma Industry
In pharmaceutical manufacturing, every detail matters, not only for operational efficiency, but for product quality, compliance, and ultimately patient safety.
Across materials, equipment, processes, and personnel, manufacturers need a reliable way to document exactly how every batch was produced.
That’s where batch records come in.
What Is A Batch Record?
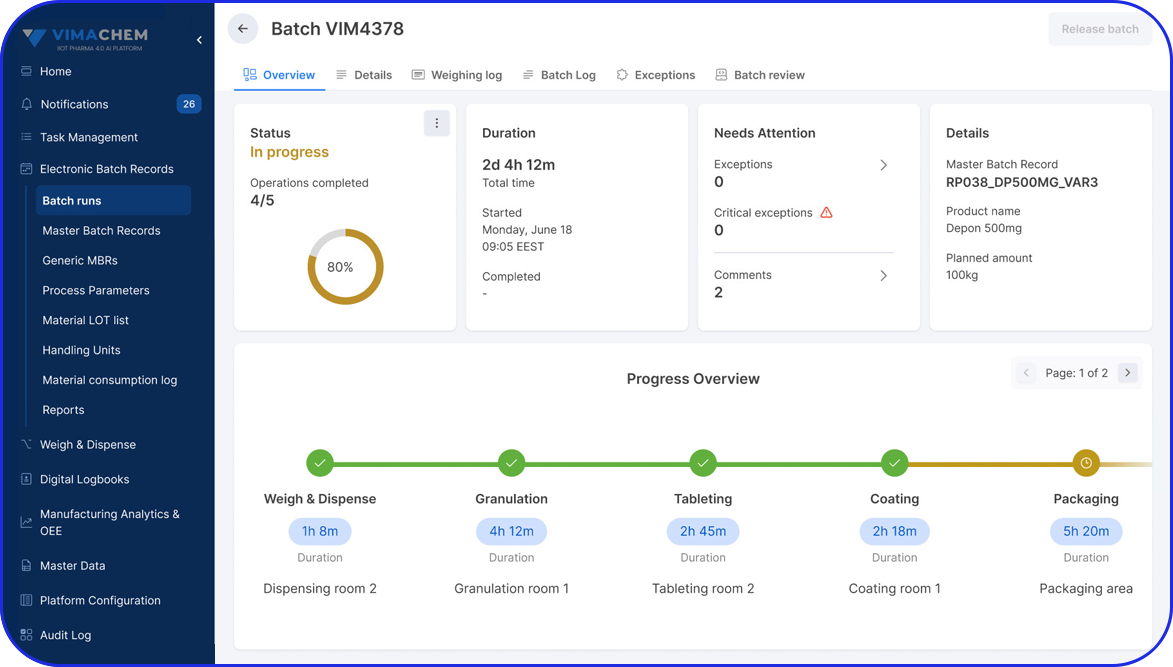
A batch record, or Batch Manufacturing Record (BMR), is a document that captures the complete manufacturing history of a pharmaceutical product batch. It includes detailed information on who performed each step, where and when activities took place, and how the batch was produced, helping ensure product quality, traceability, and compliance.
Why Are Batch Records Important In Pharma?
Batch records are indispensable tools in pharmaceutical manufacturing, as they:
- Form the foundation of pharmaceutical quality control.
- Maintain detailed records across the manufacturing process, from material usage to in-process and final quality checks.
- Ensure product quality.
- Safeguard patient safety.
- Demonstrate compliance with GMP (Good Manufacturing Practice) regulations.
- Provide a structured record of manufacturing activities for pharmaceutical manufacturers.
- Support continuous improvement and operational excellence across pharmaceutical manufacturing operations.
In pharmaceutical manufacturing, batch records serve as documented evidence of how a product was manufactured and are expected to comply with ALCOA data integrity principles.
What Does ALCOA Stand For in Pharma?
- A: Attributable – it must be clear who performed an action or created each data entry.
- L: Legible – data must be readable, permanent, and accessible.
- C: Contemporaneous – data must be recorded at the time the activity takes place.
- O: Original – the record must preserve the original data or a verified true copy.
- A: Accurate – the data must be correct, truthful, complete, and reflect what actually happened.
What Do Batch Records In Pharma Include?
Although each product’s specifics vary, the essential details of batch manufacturing records are the same across the pharmaceutical industry:
Batch Identification
A unique number, assigned to every batch produced to facilitate identification and traceability.
Composition
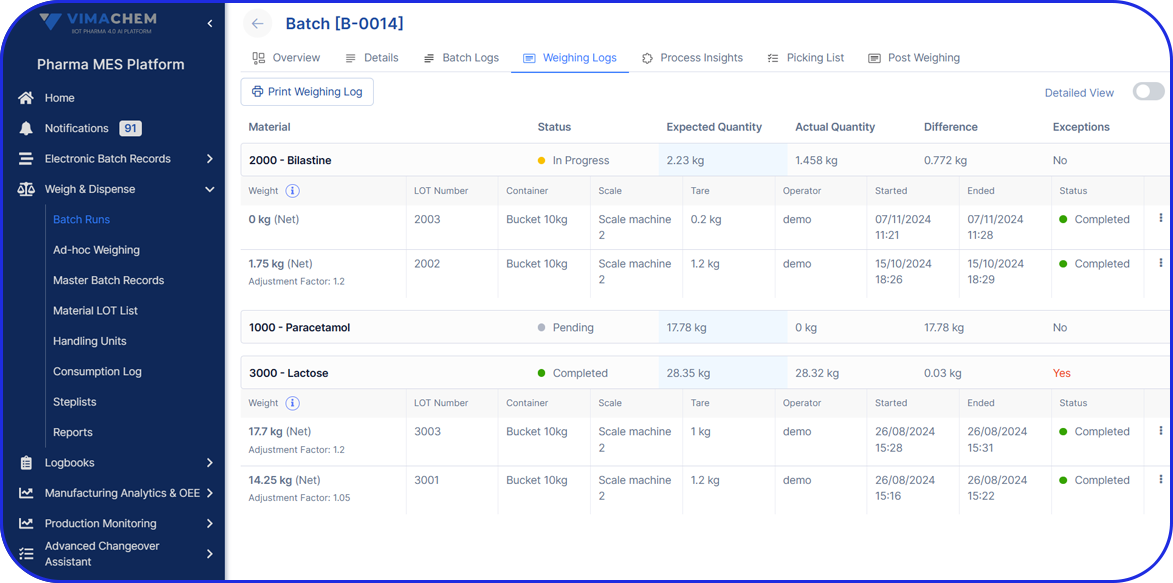
A detailed list of all ingredients and materials used in manufacturing, including quantities and relevant material identification or sourcing information.
Storage Conditions
Storage requirements for raw materials and intermediates before and during manufacturing, where applicable.
Manufacturing Procedures
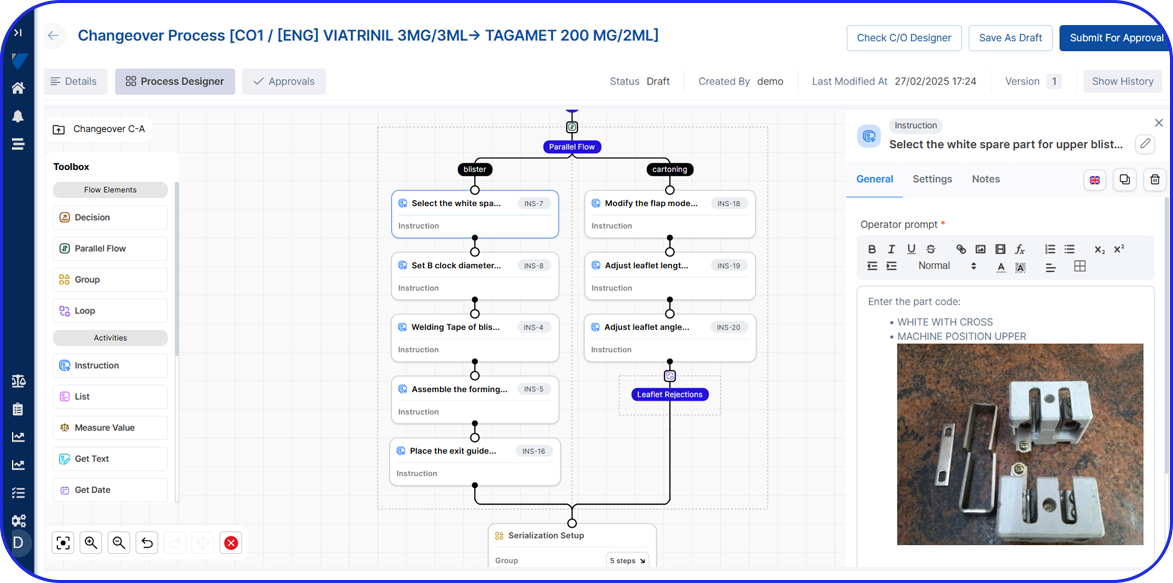
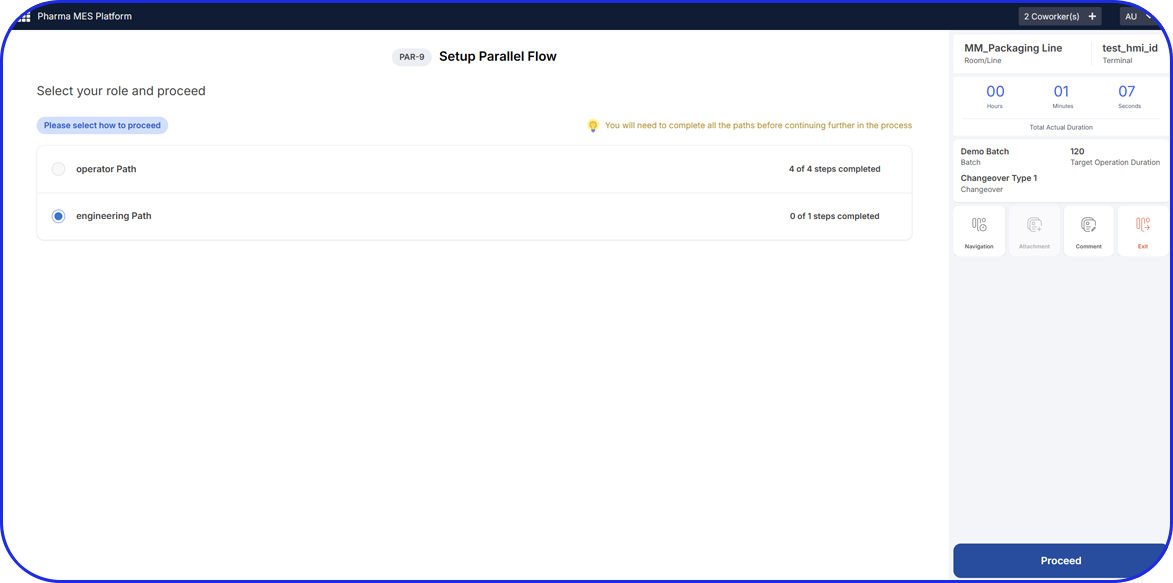
All manufacturing activities must be recorded in accordance with the approved Master Batch Record (MBR), which defines the manufacturing process. The MBR typically includes step-by-step instructions, formulae, processing parameters, equipment, and specifications.
Environmental Conditions
Information on temperature, humidity, and other environmental conditions is crucial in the pharmaceutical manufacturing process, and it’s also recorded in the batch record.
Equipment Use
Records on equipment usage, calibration, and maintenance activities, including which equipment was used, who verified it, and when it was approved for production.
Quality Control Checks
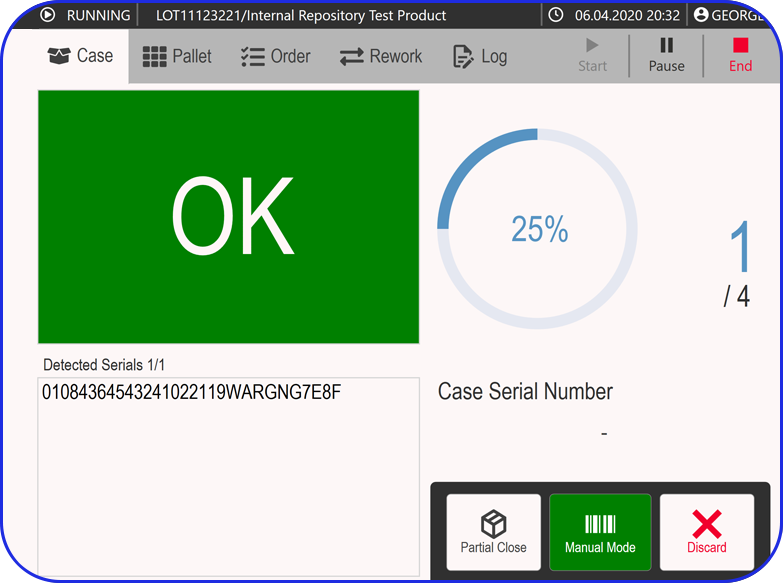
Records on all quality checks performed during and post-production.
Packaging Details
Packaging materials, labeling details, and packaging process records associated with the finished product batch.
Staff Records
Personnel involved in manufacturing activities are required to review or sign relevant batch record steps to support traceability and accountability.
Date/Time Stamps
To improve traceability, timestamps are needed for each step of the production process.
Deviation Reports
This is another essential piece of data stating any changes or deviations from standard procedures or non-conformance issues. Records also include the corrective actions taken.
So Much Data...
Managing large volumes of batch-related data efficiently is critical for maintaining compliance, traceability, and operational performance.
In the days of paper BMRs, batch record management was quite a task. And, as industry experts declare, “paper is the enemy of quality” in pharma manufacturing.
As pharmaceutical manufacturing environments become more complex and data-intensive, managing batch records digitally has become increasingly important for maintaining operational efficiency and GMP compliance.
What Is Batch Record Management In Pharma?
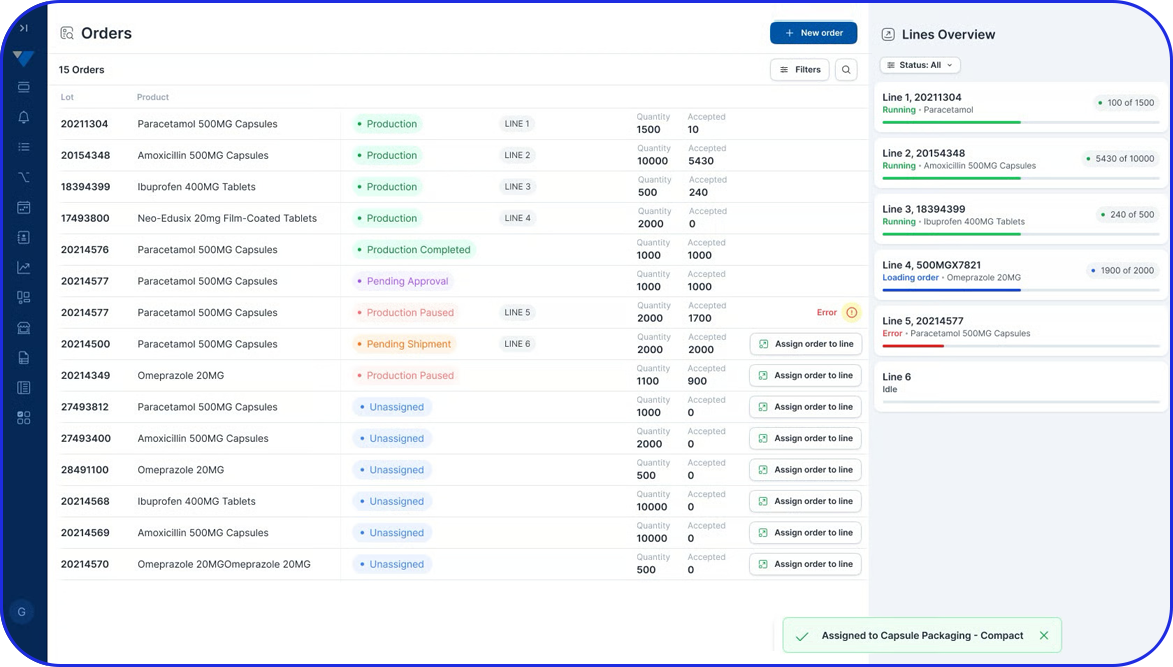
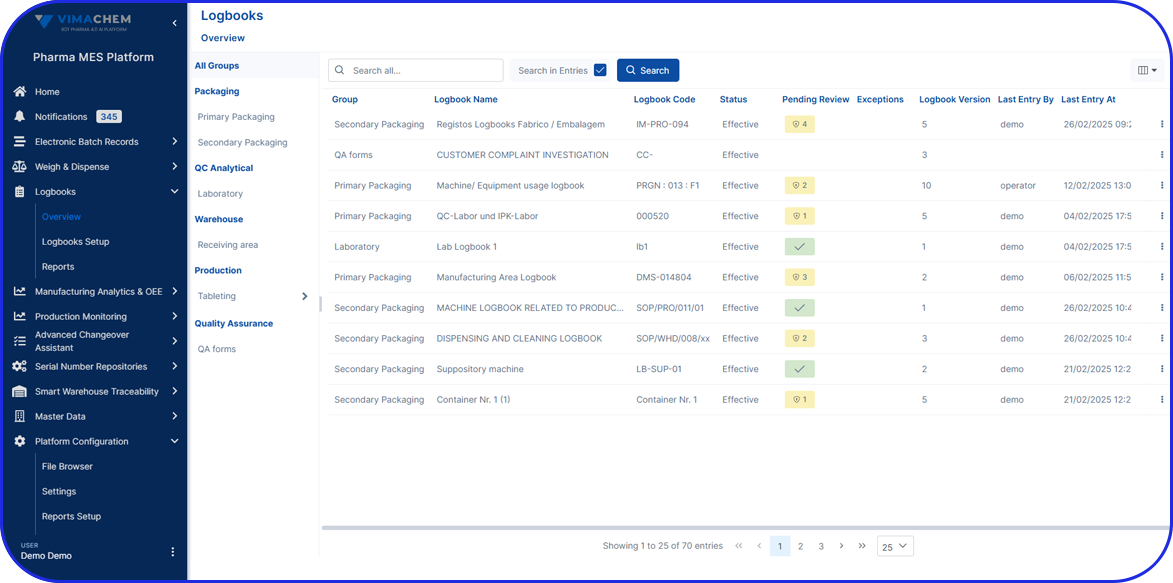
Batch record management is the entire process of creating, reviewing, approving, storing, and retrieving batch records. It’s an essential part of pharma manufacturing that helps organizations maintain compliance, improve operational consistency, and streamline manufacturing review and release processes.
In the era of Pharma 4.0, Electronic Batch Records help organizations address many of the limitations associated with paper-based batch management.
Challenges Of Paper-Based Batch Records
- Inconsistencies and data loss.
- Time-consuming manual entry.
- Scattered information across departments and systems.
- Lack of real-time visibility that hinders timely decision-making and problem-solving.
Benefits Of Electronic Batch Record Systems
Electronic batch records (EBR) address paperwork challenges in pharmaceutical production.
Modern Electronic Batch Record systems improve manufacturing execution and batch documentation through capabilities such as:
Automated Data Entry
Eliminates time-consuming, error-prone manual tasks and safeguards data integrity.
Real-Time Tracking
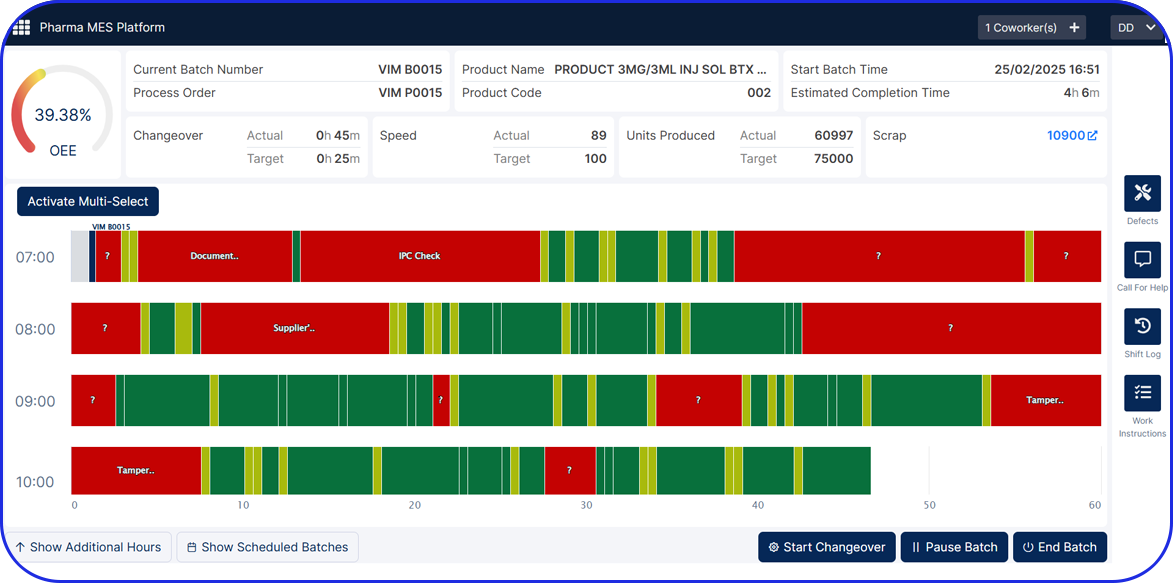
Offers complete visibility into operations and facilitates real-time, data-driven decisions.
Reduced Costs
With EBRs, production data can be securely stored and retrieved digitally, helping reduce administrative effort, manual review time, physical document storage requirements, and time-consuming record retrieval processes.
Advanced Reporting
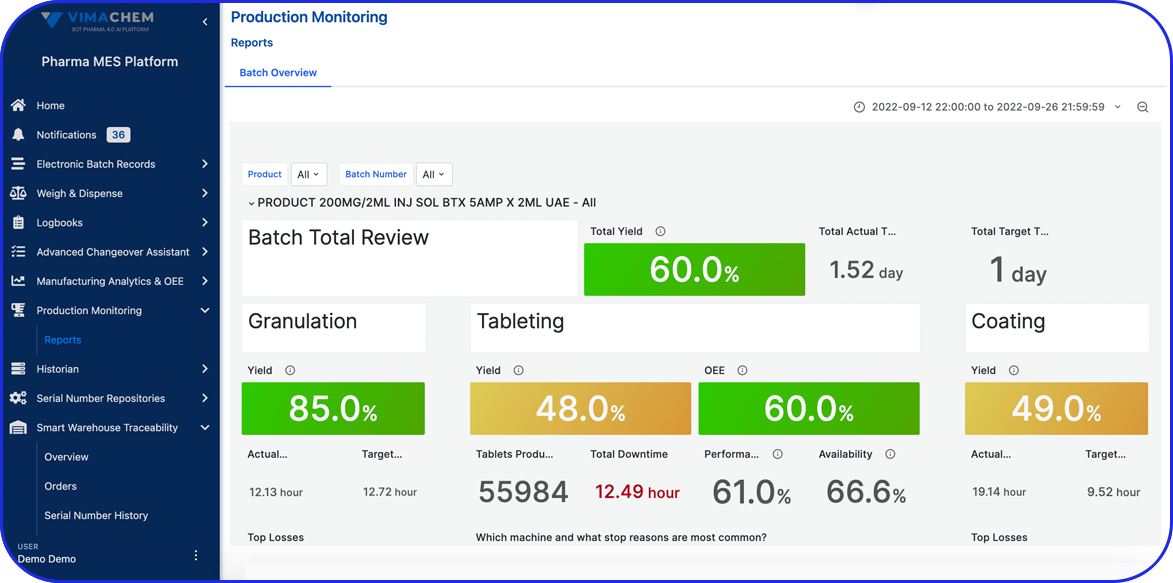
Easily generated reports and data analysis help pharma manufacturers stay on top of their production processes.
Automated Alerts
EBR systems can automatically notify teams about deviations, missing data, or process exceptions in real time.
Enhanced Traceability & Compliance
EBR systems provide a complete audit trail of all batch-related activities, making tracking and investigating issues easier. Plus, they help manufacturers adhere to compliance regulations and streamline regulatory audits.
As pharmaceutical manufacturers continue digital transformation initiatives, Electronic Batch Records are becoming an increasingly important part of compliant manufacturing operations.
The Role of Electronic Batch Records in Regulatory Compliance
Today, EBRs play an increasingly important role in supporting GMP compliance and audit readiness.
Regulatory agencies expect pharmaceutical manufacturers to maintain complete, accurate, and secure electronic records, including electronic signatures where applicable, to demonstrate that products are consistently manufactured according to approved processes and GMP requirements.
Batch records are subject to scrutiny during regulatory inspections, and any discrepancies or deficiencies can lead to warning letters, product recalls, or even plant shutdowns.
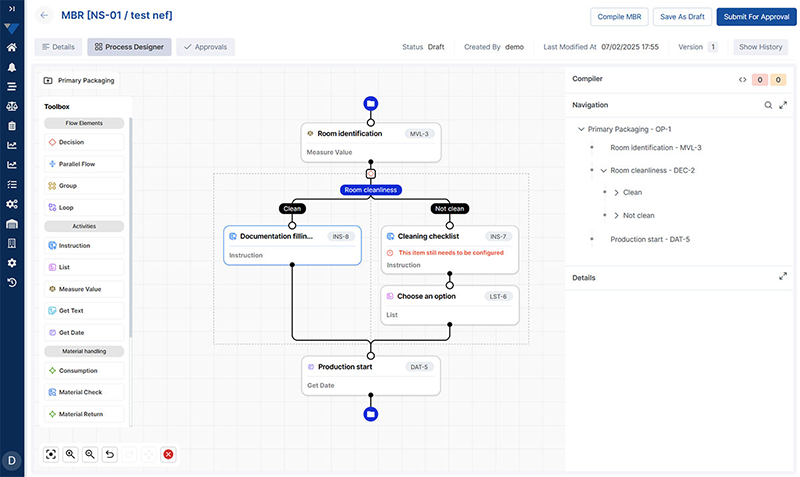
Electronic Batch Records software for Pharma
How Electronic Batch Records Improve Pharma Industry Operations - An Example
A Life Science manufacturing company decided to move from paper to an EBR system.
The upgrade’s most significant immediate impact was a 75% decrease in human error related to batch records compared to their previous hardcopy system. The reduction in manual documentation errors also supported faster review processes, improved traceability, and more consistent manufacturing execution.
Manufacturing sites reportedly generate an average of more than 1,000 deviations annually, depending on factors such as facility size, process complexity, and production changeovers.
In pharmaceutical manufacturing environments still relying heavily on manual or paper-based documentation, deviations and review delays can significantly impact operational efficiency and compliance activities.
This is one of the key reasons Electronic Batch Record systems are becoming increasingly important for improving traceability, reducing documentation errors, and supporting more consistent manufacturing execution.
Elevate Pharma Production With Cloud-Based EBRs
Vimachem’s Electronic Batch Record software is a cloud-based, pre-validated solution built exclusively for Pharma & Biopharma.
Designed to meet changing regulatory requirements, Vimachem’s EBR system integrates with IoT devices for real-time feedback on critical parameters. The result is improved data integrity, stronger traceability, streamlined batch review processes, and more consistent manufacturing execution.