What Is Batch Manufacturing Record In Pharmaceutical Industry?
What Is Batch Manufacturing Record In Pharmaceutical Industry?
Because every dose matters, precision in pharmaceutical production is critical at every stage and with every component. This tightly regulated industry, where patient safety is paramount, demands advanced manufacturing processes to ensure every drug that reaches the market meets strict quality standards.
Here is where Batch Manufacturing Records (BMRs) come into play. Having started as handwritten documents, today, they integrate gracefully with intelligent MES platform, enabling automated data capture and end-to-end evaluation for each batch, from raw materials to final product.
What Is A Batch Manufacturing Record In Pharma?
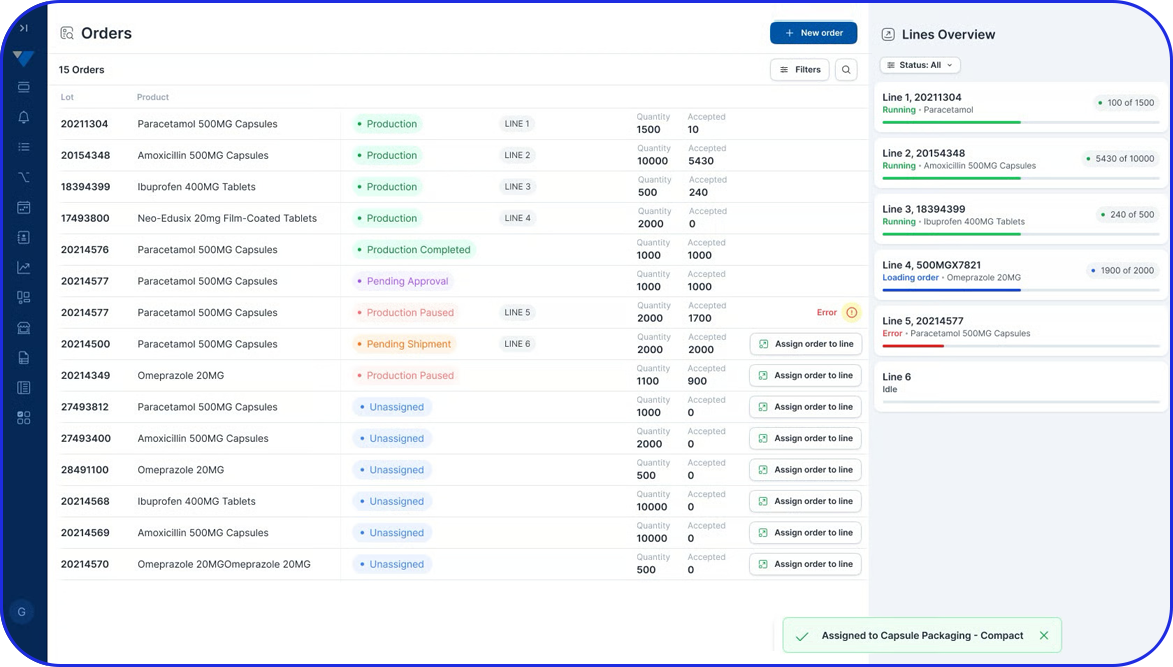
A Batch Manufacturing Record (BMR) is a comprehensive set of documents that meticulously chronicles the entire production journey of a specific product batch, recording precisely how that production occurred. For life sciences manufacturers, including those in pharmaceuticals and biotech, BMRs ensure auditability, quality control, and regulatory compliance.
In essence, a contemporaneous record of batch manufacturing activities. It captures all actions, materials, and steps involved in creating every single batch. It ultimately helps produce consistent, high-quality products that meet all safety standards, providing elaborate data and enhanced traceability.
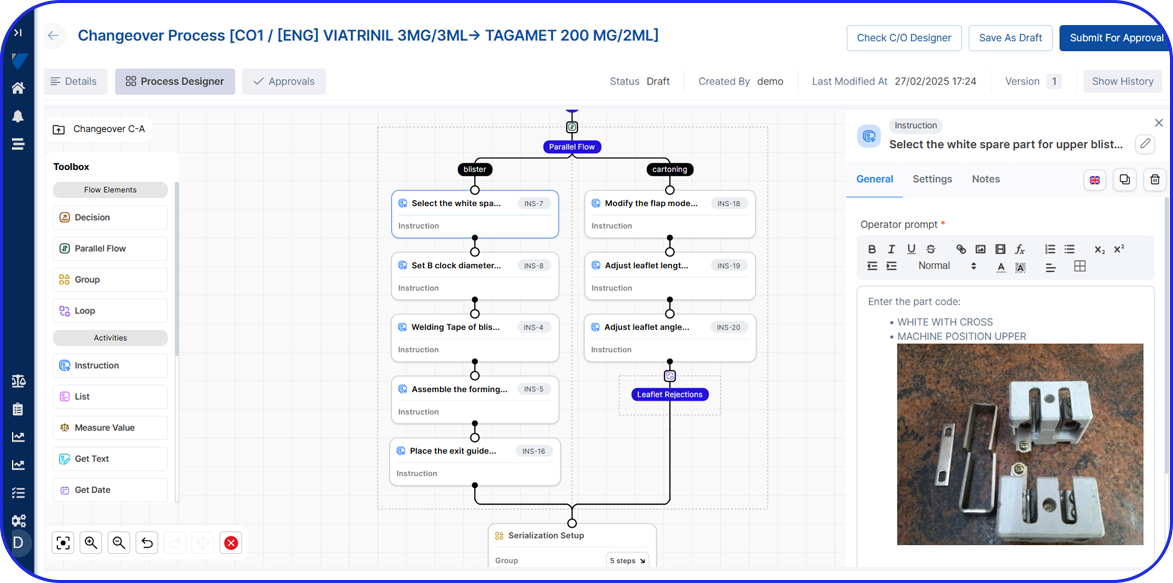
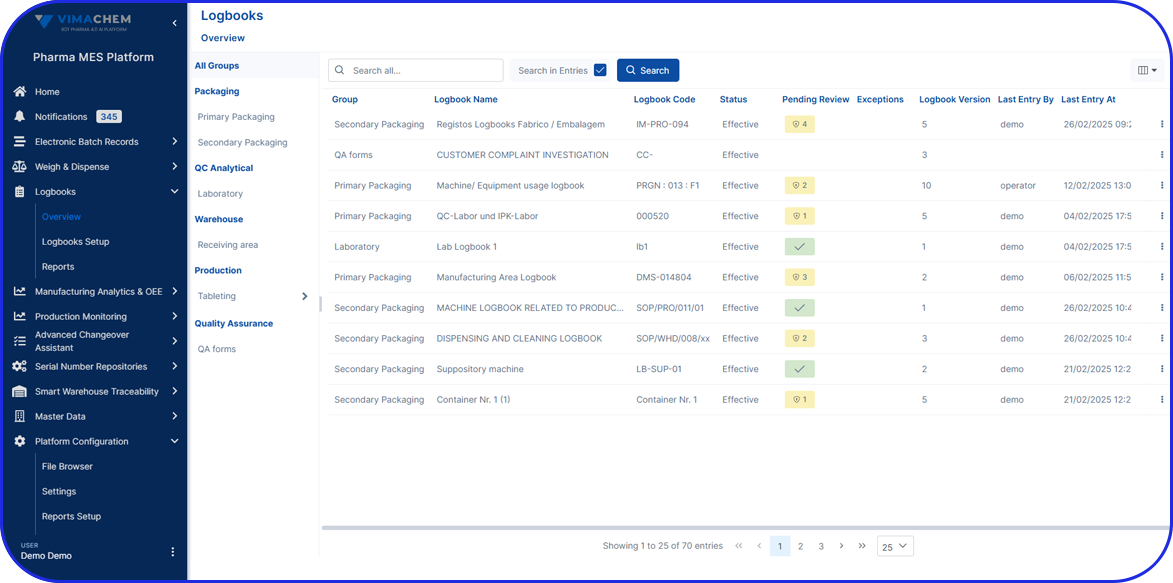
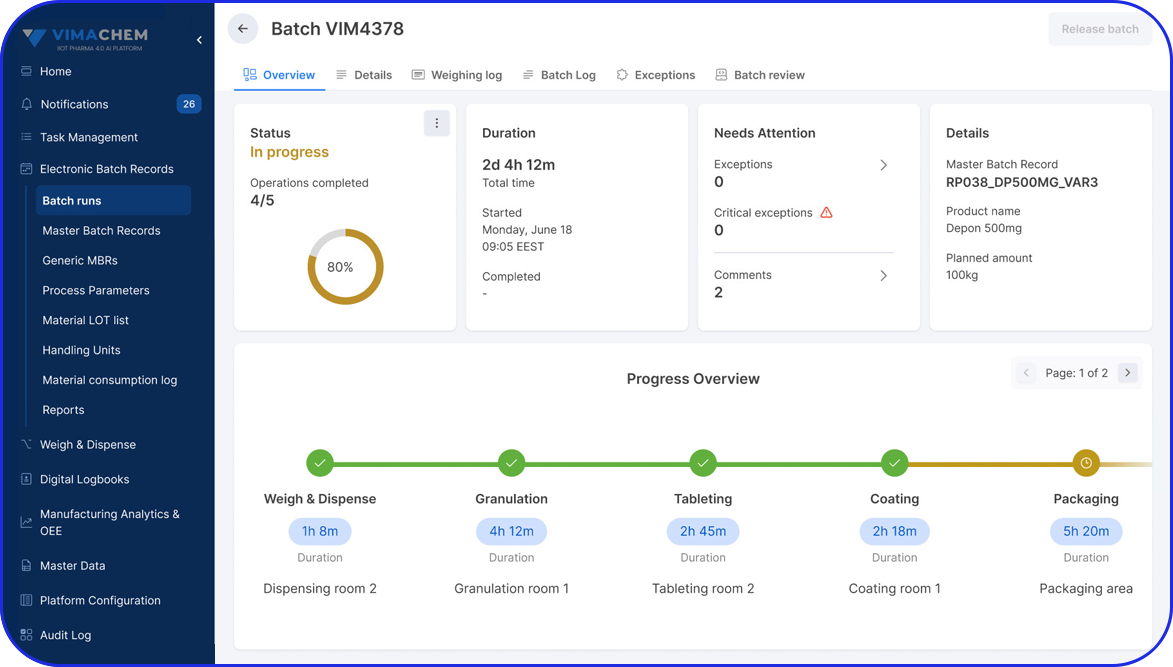
Originally maintained on paper, batch manufacturing records are increasingly implemented in electronic form as eBRs (electronic Batch Records), also sometimes referred to as electronic batch manufacturing records.
Importance Of Batch Manufacturing Records
Beyond record-keeping, BMRs in pharma safeguard patient safety, ensure quality and compliance, and improve operational efficiency in several key ways.
- Ensure Consistency & Quality by documenting each step of the manufacturing process and documenting any deviations so that each batch is produced under controlled and consistent conditions, meeting the predetermined quality standards. As an industry expert notes, “a BMR helps ensure each batch is uniform in quality and composition”.
- Facilitate Traceability & Compliance by offering a detailed trail of data from raw materials to finished products, with quick identification capabilities that help address any issues that may arise. BMRs are an indispensable tool for pharma manufacturers who must demonstrate adherence to Good Manufacturing Practices (GMP) and regulatory requirements during audits and inspections.
- Support Quality Control by documenting in-process controls, sampling procedures, and testing methods. Deviations from standard procedures are documented and investigated, supporting timely corrective actions and product quality assurance.
- Enhance Patient Safety thorough documentation of all manufacturing steps supports assurance that the final product meets established safety and quality requirements.
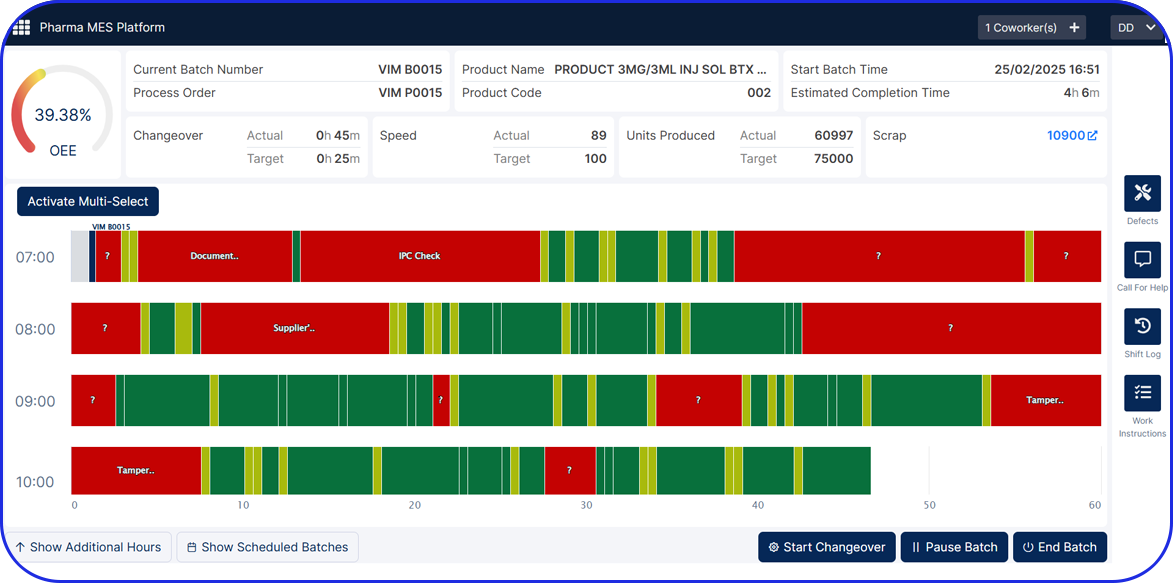
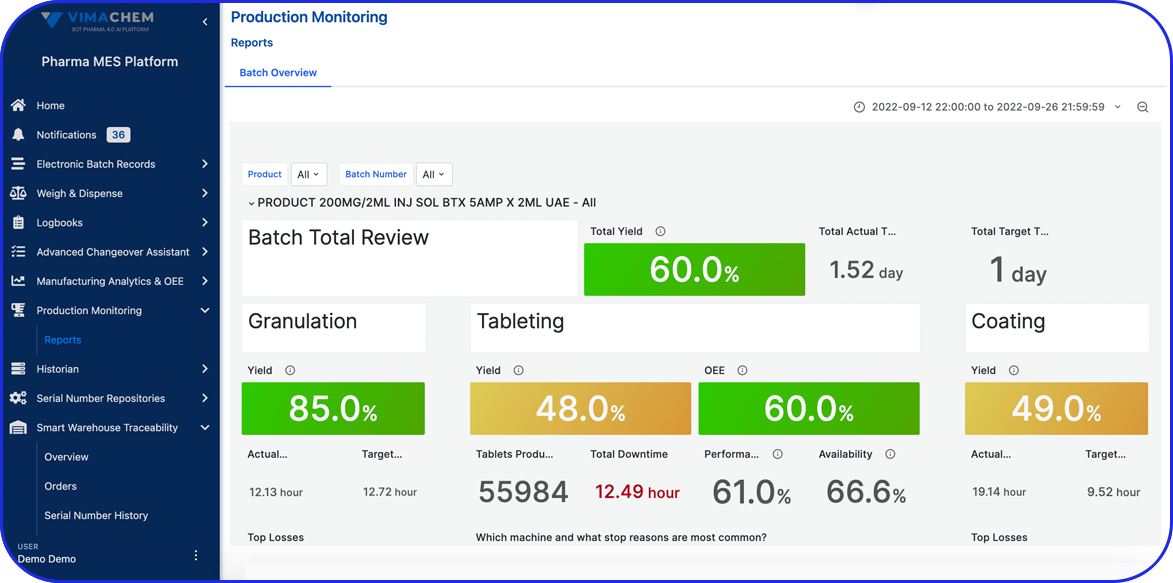
- Improve Efficiency – a well-documented BMR allows manufacturers to spot inefficiencies or problems in the production process, making it easier to fix issues and streamline future batches. Specifically, electronic Batch Manufacturing records enable automated calculations, electronic signatures, and document retrieval, significantly improving overall efficiency.
What Is Included In A Batch Manufacturing Record?
BMRs include a wide array of information to ensure a complete and verifiable history of a product batch. While drug production processes differ, here’s a batch manufacturing record example of what’s generally included:
- Title Page: Document title (Batch Manufacturing Record), Product Name, Batch Number, Batch Size, Manufacturing Date, Expiry Date, and an internal BMR Number.
- Table of Contents: A detailed list of all sections for easy navigation.
- Product Details: Dosage form, strength, manufacturing site, license number, and a reference to the approved Master Batch Record or Master Production Instruction.
- Batch Formula: Lists of active pharmaceutical ingredients (APIs) and excipients, along with their specific quantities.
- Manufacturing Process Overview: A visual flow diagram and critical process parameters (e.g., temperature, pressure) that must be controlled.
- Equipment and Facilities: Identification numbers, calibration status, and cleaning/maintenance records for all equipment used.
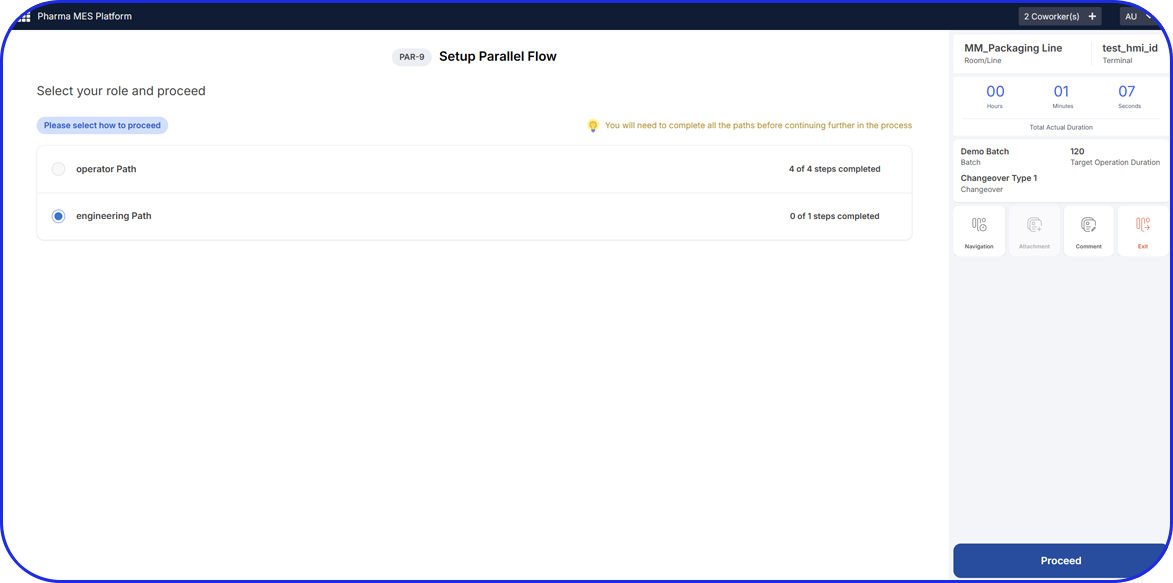
- Manufacturing Instructions: Detailed, step-by-step procedures for each stage, such as weighing, mixing, granulation, drying, compression, coating, and packaging.
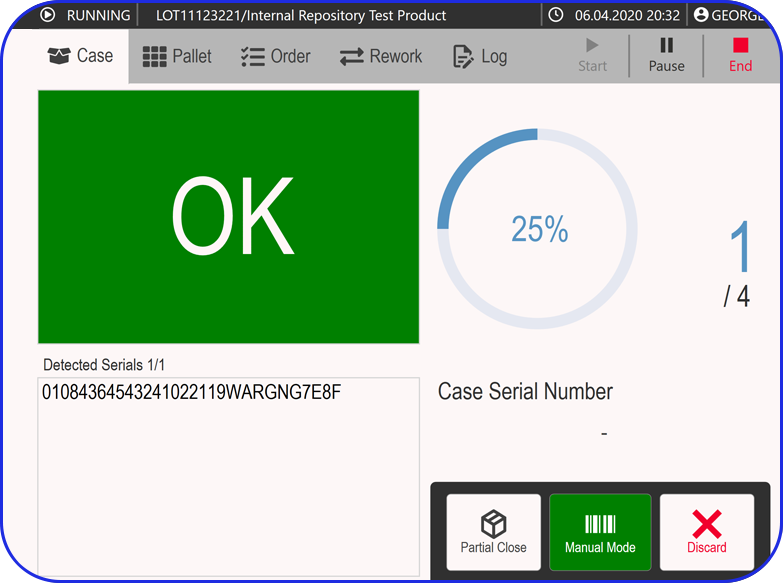
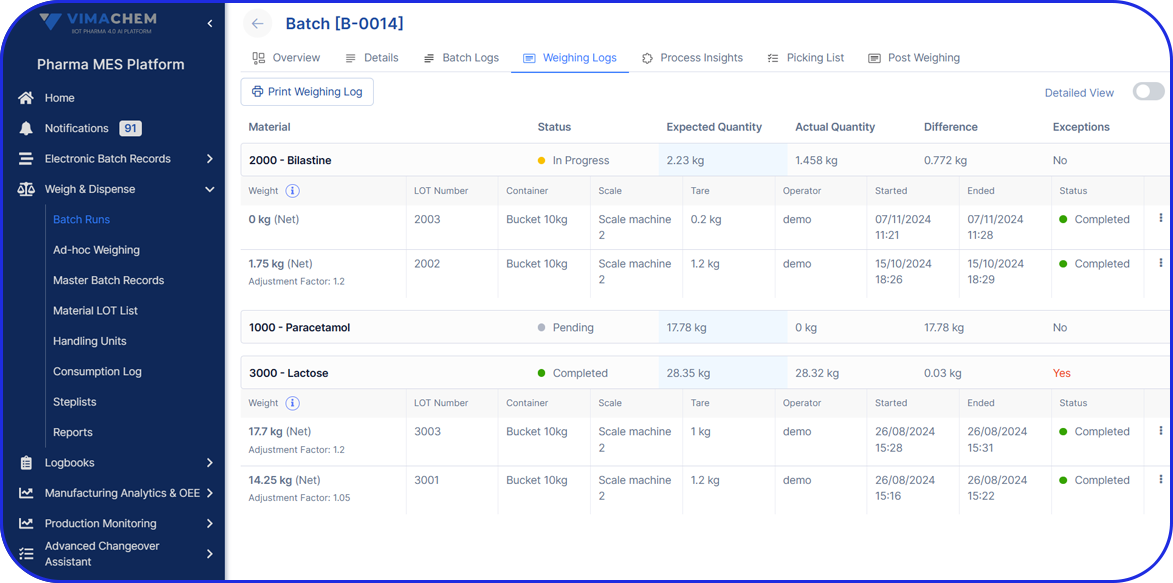
- In-Process Controls: Sampling and testing procedures, specifications, acceptance criteria, and actual test results for samples taken during manufacturing.
- Packaging Instructions: Specifications for packaging materials, detailed operations, labeling procedures, and batch coding for traceability.
- Quality Control: Detailed tests and specifications for the final product, final product testing results and related quality documentation, environmental monitoring, and final product testing results.
- Deviation Management: Procedures for recording any deviations from the approved process, including nature, cause, corrective actions, and preventive actions (CAPA).
- Personnel & Training: Names and roles of personnel involved, with signatures or electronic sign-offs verifying completion of each step, including dual verification where required.
- Approval & Release: Quality Assurance (QA) approval, final batch release sign-off, and signatures of authorized personnel
- Appendices: References to relevant Standard Operating Procedures (SOPs), raw data, calculations, analytical test reports, and equipment calibration certificates.
- Record of Revisions or Changes: Documentation of any approved revisions or changes recorded during batch execution, including dates, details, and authorization.
Types Of Batch Manufacturing Records
While the BMR itself is the core record of a batch production, the manufacturing process often involves a hierarchy of related records:
- Master Batch Records (MBRs): The predefined documents that serve as a production blueprint. They outline the standard manufacturing process and act as a guide for producing specific batches.
- Quality Control Records: Documentation of sampling, testing, and results associated with the batch.
- Master Packaging Records (MPRs) / Batch Packaging Records (BPRs): For pharmaceutical products that involve packaging bulk finished goods into final presentation (e.g., tablets into bottles or blisters), dedicated packaging records are used. The MPR defines the approved packaging instructions, while the BPR documents how a specific packaging run was executed, acting as the packaging equivalent of MBRs and BMRs.
- Validation Batch Records: These records define validation instructions and document execution results of product validation processes.
Regulatory Requirements For Manufacturing Batch Records In Pharma
In short, BMRs must demonstrate that all drug products are manufactured in accordance with specified standards. To do so, they should include information about raw materials, process steps, and tests performed to verify quality, all of which must be verified and signed by a quality assurance representative.
Here’s an overview of relevant regulatory requirements:
- FDA Guidelines (U.S. Food and Drug Administration): The FDA requires companies to create BMRs for drug products, to ensure quality and safety. Under 21 CFR Part 211.188, BMRs must comprehensively document each significant step, including dates, major equipment used, quantities of components, in-process and laboratory control results, and investigations into any deviations. The FDA uses BMRs to verify that health and safety procedures were followed, quality control testing was performed, and ingredients were handled properly.
- EMA Guidelines (European Medicines Agency): The EMA mandates that BMRs include data confirming that each batch was produced and checked in compliance with the marketing authorization and GMP, as articulated in Chapter 4 of EudraLex Volume 4.
- WHO Guidelines: To ensure drug integrity and traceability globally, the World Health Organization provides comprehensive guidelines for the production of pharmaceuticals that cover identification, product information, manufacturing instructions, raw materials, in-process controls, packaging and labeling, quality control testing, deviation and corrective actions, and signatures and approvals. They also recognize the use of electronic Batch Records and emphasize data integrity, retention, and auditability for efficiency and emphasize data integrity, retention, and auditing.
Auditing Requirements
This is a core process that reviews and verifies the accuracy and completeness of the records associated with the production of a single batch.
Components that are reviewed include:
- Raw materials and ingredients used, including test results and certificates of analysis.
- Production processes, parameters, instrument readings, equipment settings, and operator documentation.
- Finished product specifications and test results.
- Records associated with product release, such as certificates of analysis and sterility testing.
- Potential changes to the batch production process.
Beyond standard BMRs, electronic Batch Records help streamline audits and clearly document compliance for regulatory review.
Accelerate ROI by digitizing your manufacturing with Vimachem MES.
By integrating seamlessly with your ERP and MES, it transforms traditional BMRs into automated, searchable, and traceable digital records, enabling faster batch release and stronger quality control.