Pharma MES USA 2026
Join Vimachem at Pharma MES USA – The Leading MES Conference for Pharma and Biotech Manufacturing in the United States.
Dedicated exclusively to Manufacturing Execution Systems (MES) in pharmaceutical and biotech production, the annual show brings together MES leaders, and digital manufacturing professionals to explore how MES drives operational excellence, compliance, and Pharma 4.0 transformation.
Gain practical insights into building connected shop floors, leveraging MES for predictive quality, and achieving next-level plant productivity. oin us in Boston to network with peers, benchmark best practices, and define your roadmap to digital transformation and manufacturing excellence.

March 23 - 24, 2026
Encore Boston Harbor

Join Vimachem
At Pharma MES USA
Connect with Vimachem at the event





Join the Session
On Monday at 14:10, join our customer Steven Kinney, Director at Sharp Sterile Manufacturing, and Vimachem CEO Alex Vidras to learn how Sharp Sterile is "Standardizing Manufacturing Analytics & OEE Solution Across Sites".
Join the Session
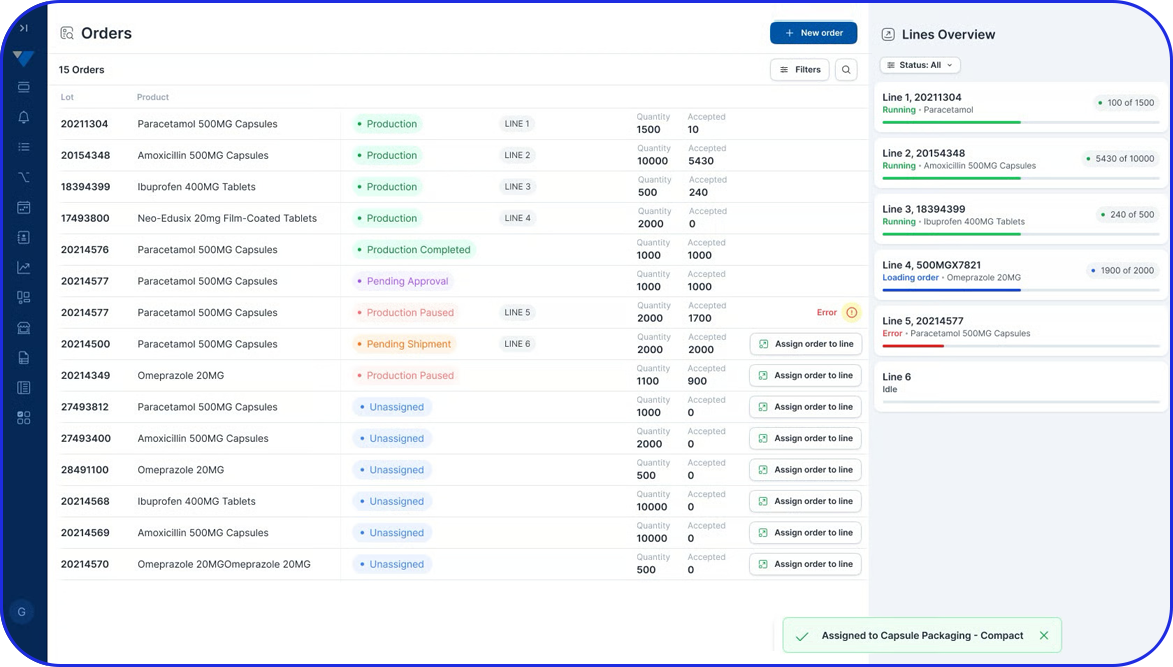
Purpose-built for the life sciences industry, the Vimachem MES modular platform has been deployed across 200+ pharma sites globally, helping manufacturers boost process efficiency, enhance product quality, and reduce compliance risk and lead times.
Smart, flexible, and built with the pharma operator in mind, the Vimachem Pharma MES leverages Artificial Intelligence (AI), the Industrial Internet of Things (IIoT), and cloud computing to enable rapid, scalable digital transformation. Its modular architecture allows for deployment as a standalone solution or full integration with enterprise systems like ERP, CMMS, QMS, and LIMS, enabling implementation in weeks and delivering fast time to value.
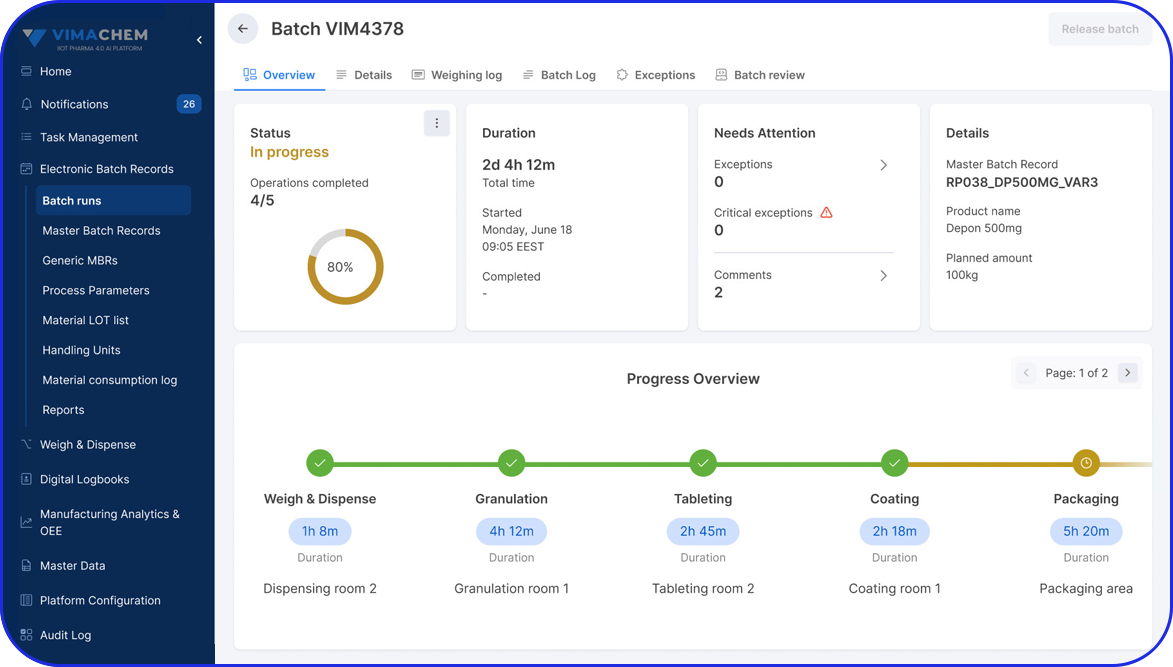
- eBR: AI-powered batch digitization with a flexible designer and review by exception.
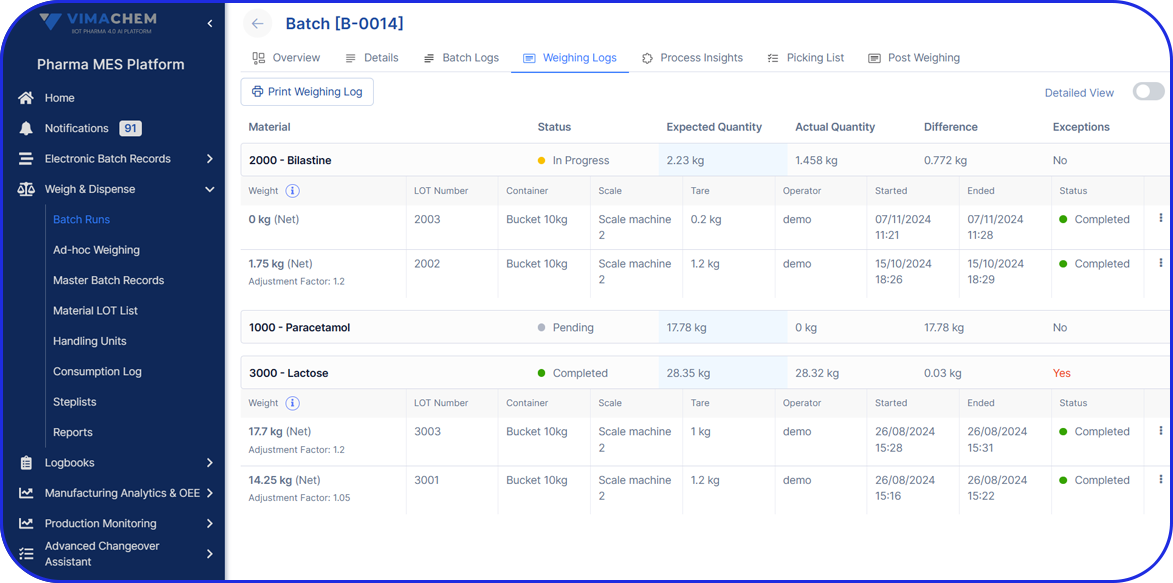
- Weigh&Dispense: Native connectivity with all major pharma/biopharma industrial scales.
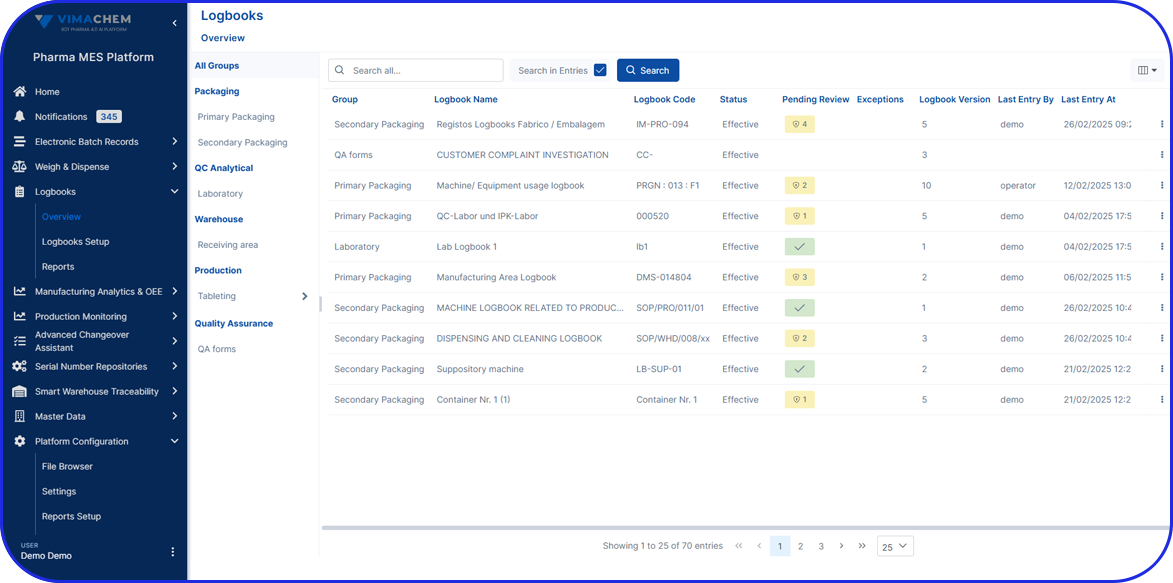
- eLogbooks: AI-driven digital logbooks with room status and workflow management.
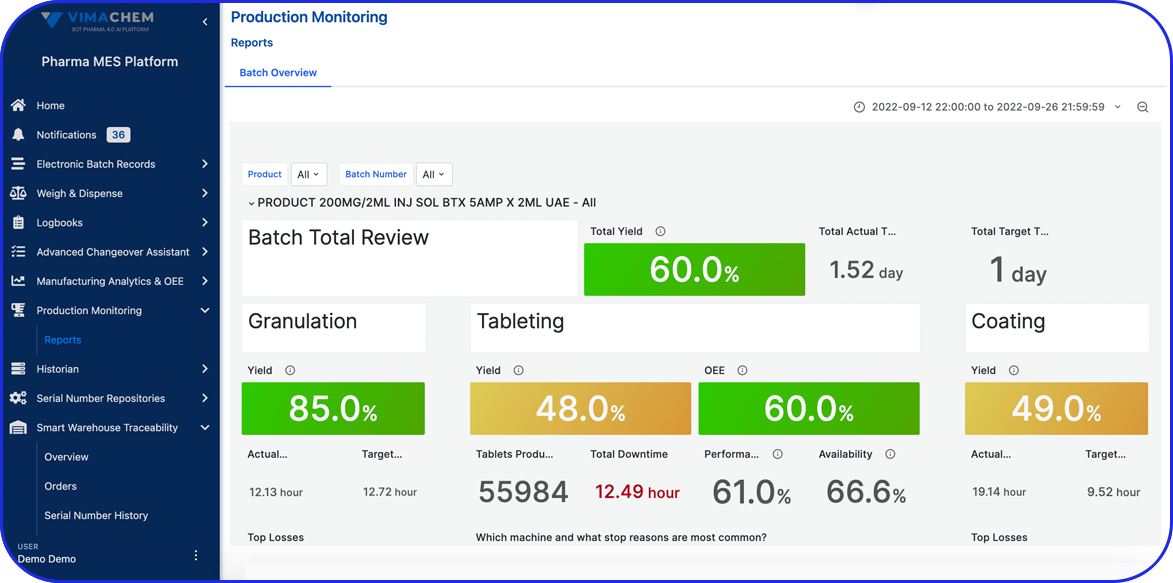
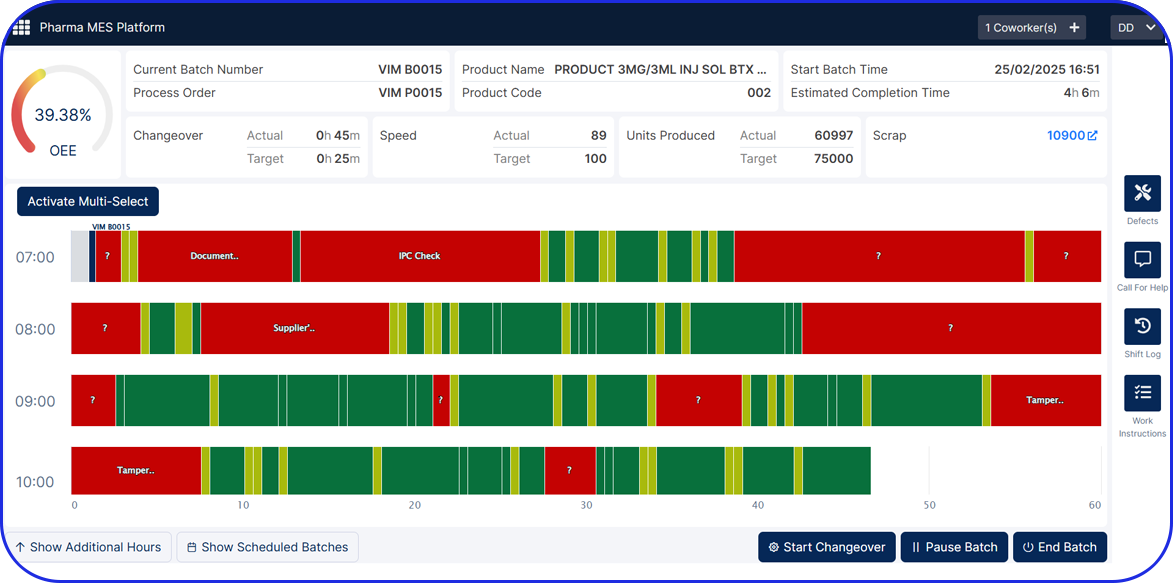
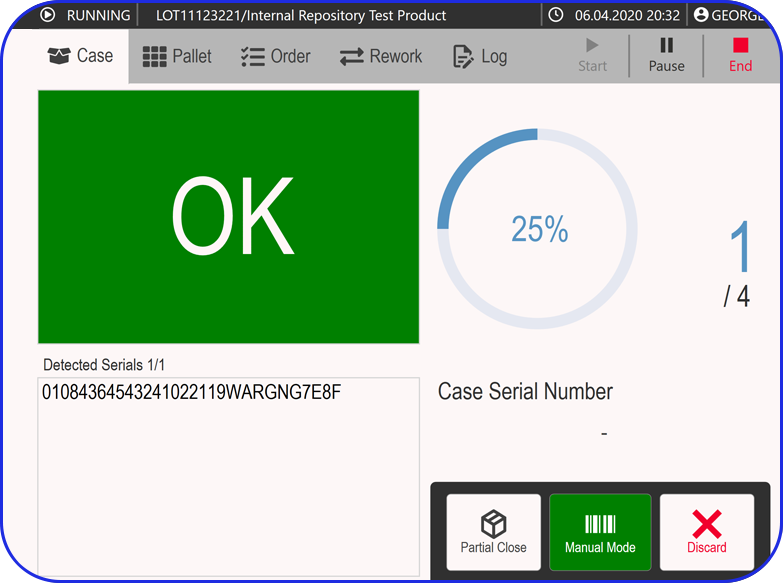
- Manufacturing Analytics & OEE (MA&OEE): Real-time insights to increase efficiency, reduce downtime, and optimize performance.
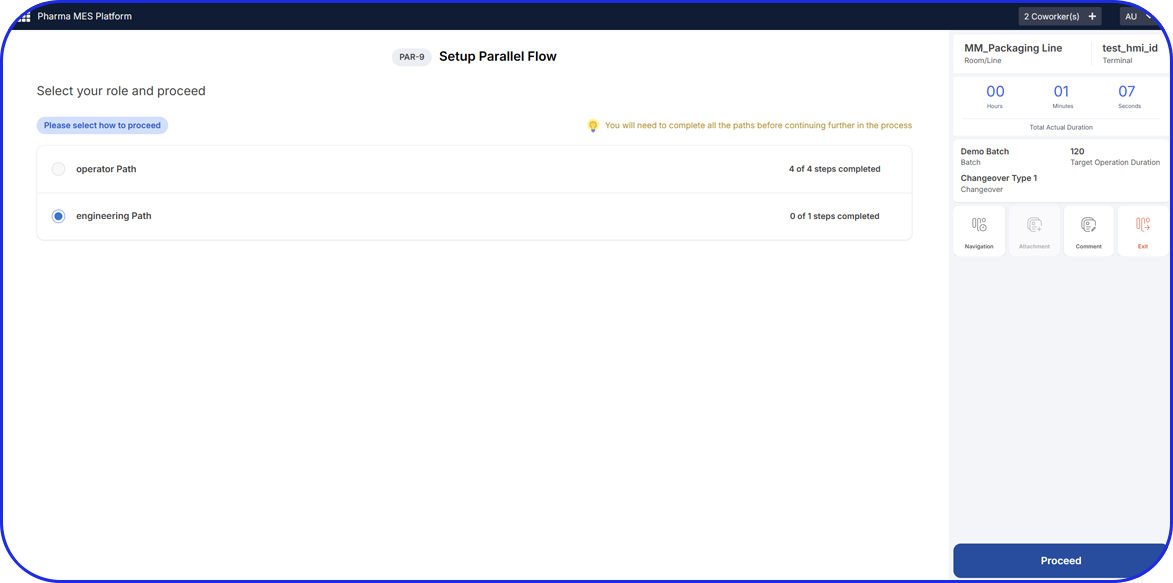
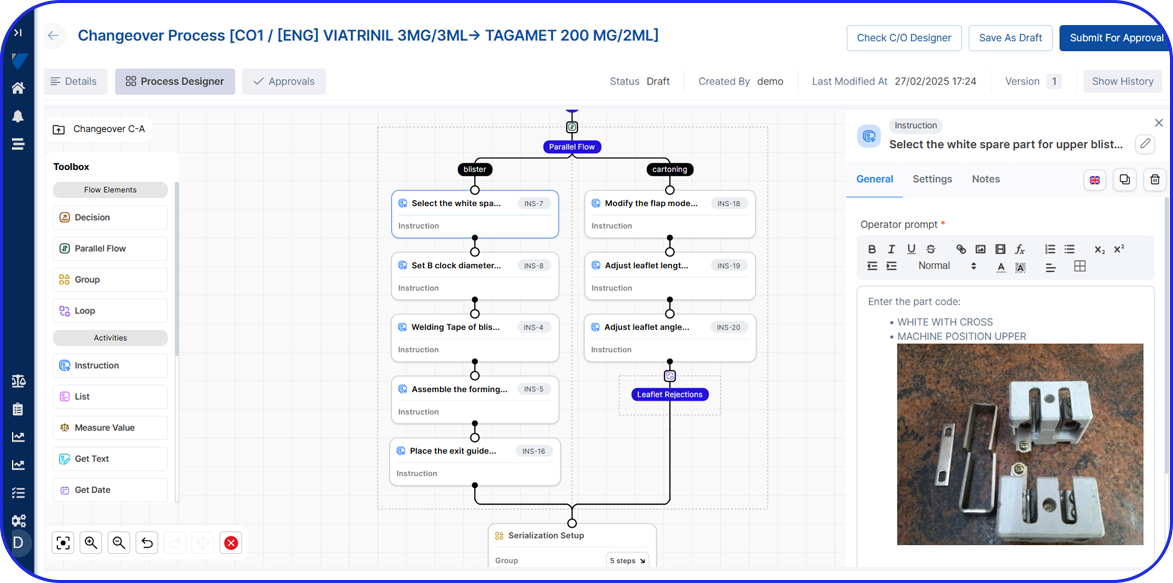
- Digital Work Instructions (DWI): Step-by-step instructions for changeover, cleaning, and in-process control (IPC).
- Serialization Manager: Level 3&4 serialization with pre-validated interfaces to major Level 2 and Level 4/5 systems.
- IIoT Edge Machine Connectivity Module.
To learn more explore our website
GxP Compliance





Until Pharma MES Europe
- 00
Days
- :
- 00
Hours
- :
- 00
Minutes
- :
- 00
Seconds
Meet our team and Explore how Vimachem’s MES can help you ensure digital compliance, enhance product quality and accelerate digital transformation.