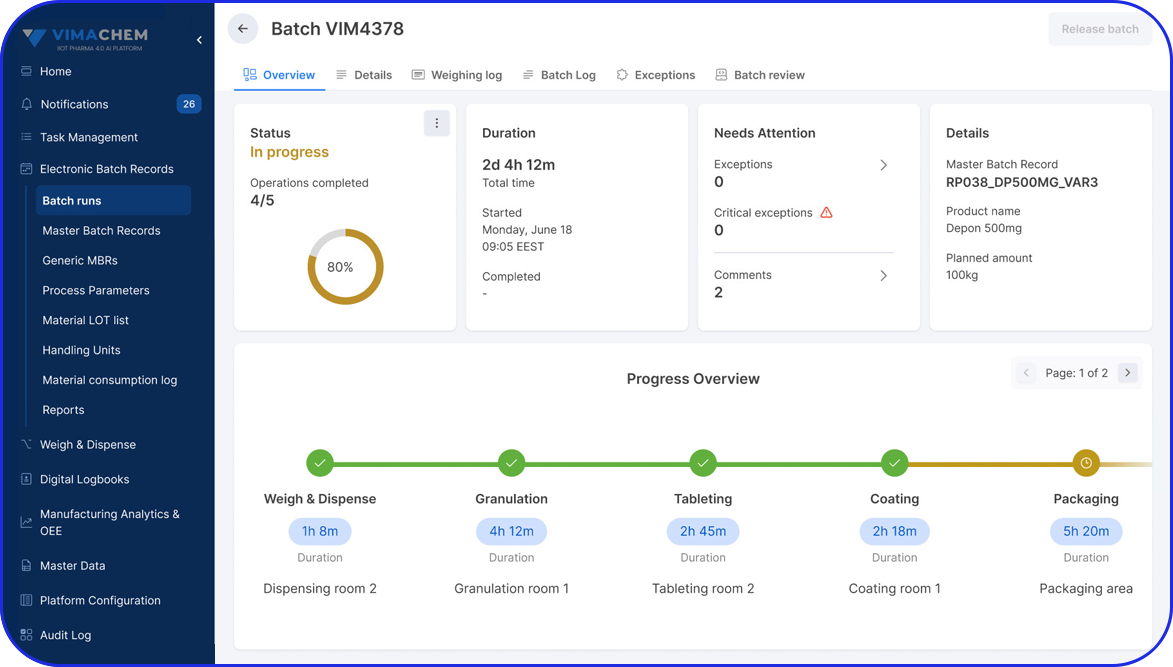
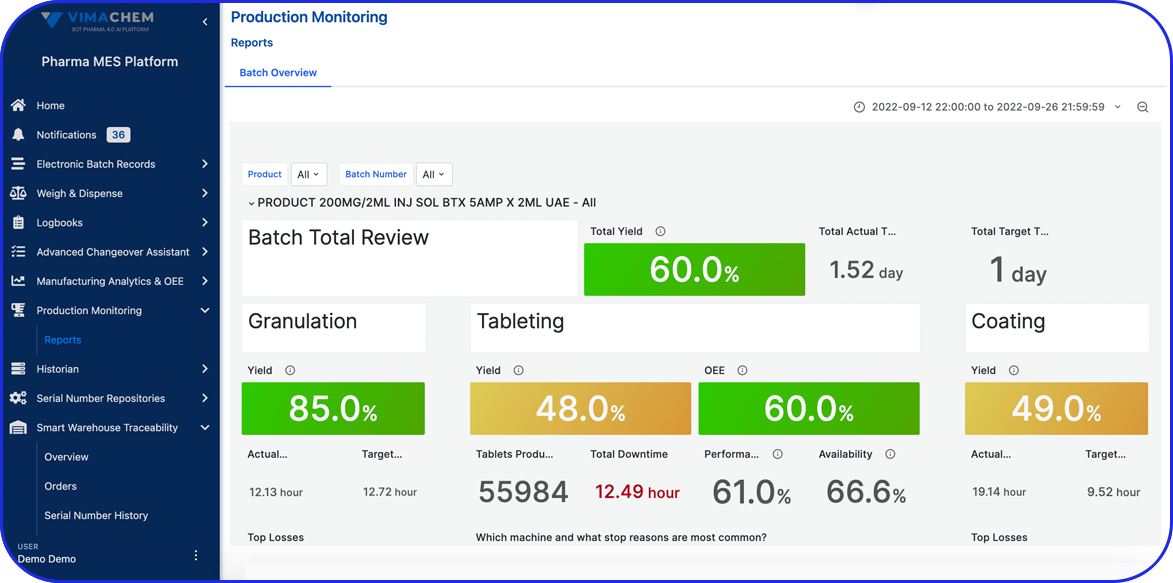
Egypt Pharma Track & Trace: What’s Coming in 2026 and How Vimachem Can Help You Comply
Egypt Pharma Track & Trace: What’s Coming in 2026 and How Vimachem Can Help You Comply

Saud Khan
Senior IT Serialization Consultant
Egypt is moving quickly toward full pharmaceutical traceability with key deadlines in February and August 2026. This represents significant momentum for Egypt to achieve more visibility into the pharmaceutical supply chain and enhanced anti-counterfeiting protection while ensuring better control over pharmaceutical products. The message for partners selling into Egypt, whether a local manufacturer, importer, MAH, CMO, 3PL, or distributor, is simple: “Get ready” before the penalties begin.
Regulatory Timeline at a Glance
The legal foundation is EDA Decree No. 475 of 2025, which introduces the national Track & Trace system and sets clear enforcement milestones: finished imported medicines must comply from 1 February 2026, and locally produced, filled, or packed products must comply from 1 August 2026. After those dates, products cannot be legally circulated unless they are included in the national traceability system. Products already on the market before these dates may continue circulating until consumption or expiry, but products manufactured or imported after the deadline must be included in the system.
What This Means for Egypt
Egypt’s approach is not just about printing a two-dimensional barcode. It combines government-controlled serial number issuance, standardized GS1-based serialization on packs, aggregation for logistics units, and event reporting to the national system.
At pack level, companies are expected to apply a GS1 DataMatrix carrying the core identifiers (GTIN, serial/UID, expiry date, batch). A key differentiator is that the UID (serial number) is issued by the national Track & Trace system: companies must request UID ranges in advance (either through integration APIs or through a manual portal file download), and self-generated serials are not accepted.
To make traceability operational beyond packaging, Egypt also expects aggregation and digital reporting of lifecycle events such as commissioning, aggregation, shipping, receiving, and dispensing. In other words, compliance is achieved when packaging and warehouse operations reliably generate clean traceability data.
Who Should Act Now?
As with every modern serialization rollout, the scope extends well beyond manufacturers:
- MAHs supplying the Egyptian market.
- Local manufacturers and packaging partners (including local and contract packaging).
- Importers and CMOs packaging abroad for Egypt.
- Distributors and 3PL warehouses operating in Egypt.
- Dispensing points (pharmacies/hospitals) as the downstream endpoint of the traceability chain.
The practical reality is simple: if any link in the chain cannot handle serialized and aggregated stock correctly, data integrity breaks down and operational risks rise.
Why Early Preparation Matters
Egypt’s model has a higher compliance complexity in three distinct ways that make early planning essential. First, companies cannot create their own serial numbers. Because UIDs are part of the national system, each company must be ready to request UIDs, assign those UIDs to packaging orders, complete UID reconciliation, and maintain audit trails.
Second, compliance is measured through reporting and operational events, not only packaging capability. If commissioning, aggregation, shipping, and receiving processes aren’t stable, businesses face avoidable delays, exceptions, and market release pressure.
Third, warehouses become serialization-critical. Once aggregation is in play, warehouses must preserve parent–child relationships during receiving, put-away, picking, shipping, returns, and recalls otherwise traceability becomes theoretical rather than operational.
Why You Should Choose Vimachem as Your Compliance Partner
At Vimachem, our key differentiator is not just compliance but flexibility, scalability, and operational excellence.
Direct Connectivity to the National Egypt Track & Trace System
A central element of Egypt’s Track & Trace regulation is the nationally issued serial numbers (UIDs). Unlike other markets, companies cannot generate their own serial numbers. All UIDs must be obtained directly from the Egyptian Track & Trace system. Vimachem’s primary offering enables MAHs to connect seamlessly to the national system through our Level 3/4 Serialization Site Manager. Vimachem manages the complete UID lifecycle, including requesting, receiving, allocating, reconciling, and reporting UIDs, while ensuring event data and compliance reporting are fully aligned with regulatory requirements.
Modular, Flexible, and Line-Agnostic
Vimachem’s platform is modular by design, enabling companies to deploy only what they need, when they need it. The Vimachem Serialization Site Manager Level 3/4 solution is L2-agnostic, giving our customers the flexibility to select the most appropriate Level 2 solution for each packaging line from high-speed automated lines to low-speed or semi-automatic lines or even fully manual serialization and aggregation scenarios. This flexibility avoids overengineering and significantly reduces long-term cost and vendor lock-in. With 50+ customers globally using various L2s across the site, we have proven that flexibility and avoidance of vendor lock-in are the most secure and cost-efficient way to initiate and scale your pharmaceutical serialization operations.
Built for Zero Downtime Operations
Serialization should never compromise supply continuity. Vimachem’s solutions are designed to go live with no production downtime, supporting parallel rollout, phased go-lives, and controlled validation strategies. This allows companies to remain fully operational while upgrading their serialization and reporting capabilities.
The Vimachem Serialization Modules
We provide pharmaceutical companies with modular and regulation-ready serialization solutions that support pharmaceutical companies across packaging, warehousing, and distribution.
Our portfolio is designed to scale with evolving global requirements, including emerging markets such as Egypt:
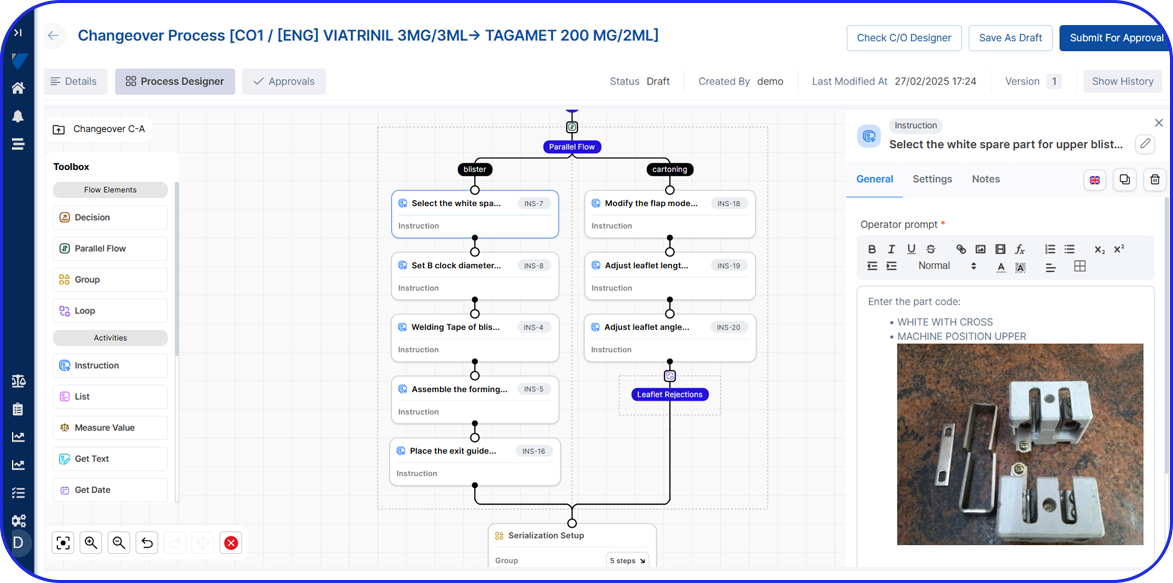
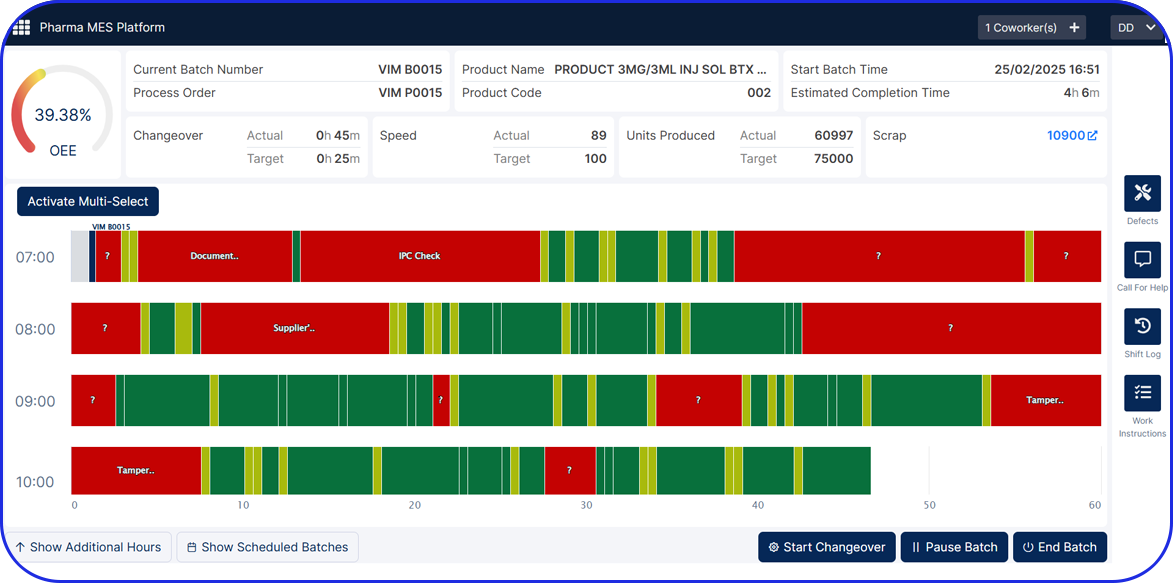
Vimachem Level 3 / Level 4 Serialization Site Manager (SSM)
Egypt’s centrally issued UID model makes Level 3 the heart of compliance. Vimachem Level 3/4 SSM is designed to be the central hub between packaging lines (Level 2) and enterprise and regulatory connectivity, reducing compliance risk and go-live pressure while enabling companies to operationalize Egypt’s requirements with control and auditability. With Vimachem Level 3/4 SSM, companies can:
- Orchestrate national UID supply (request, receive, manage, allocate, reconcile) so packaging lines always use valid UID ranges and exceptions are traceable.
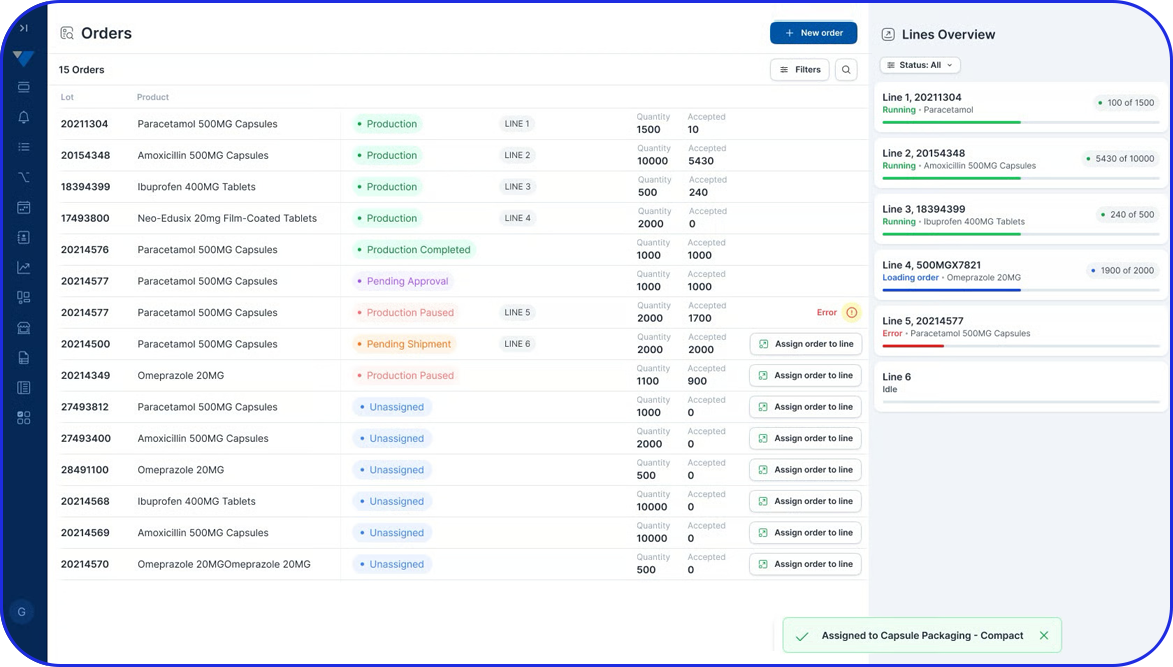
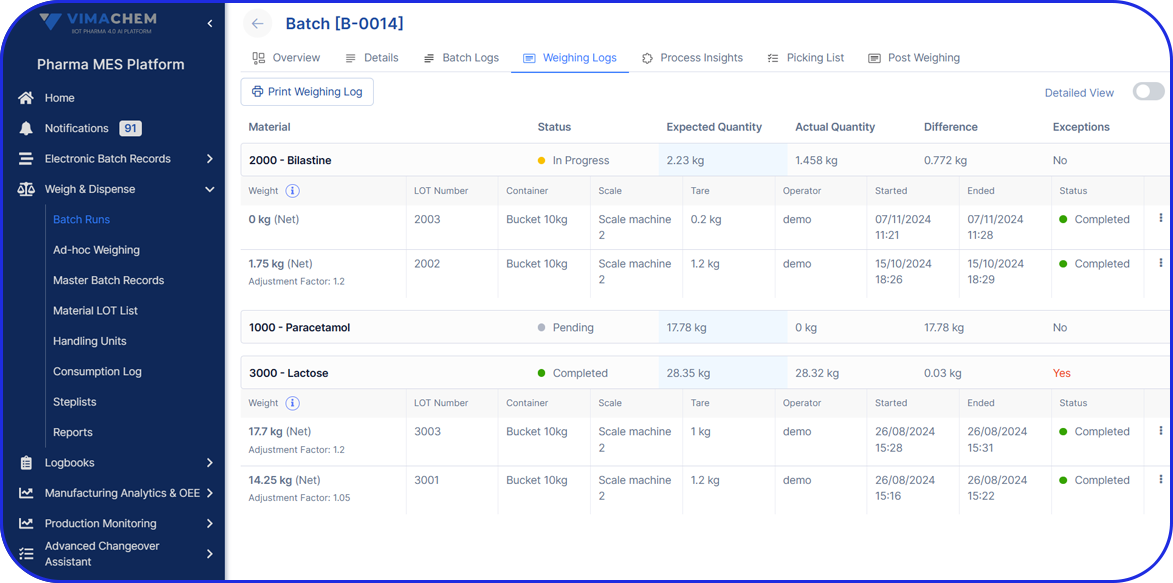
- Manage commissioning and aggregation at site level, maintaining clean serialization and aggregation that remains consistent from line to warehouse.
- Support compliance reporting workflows aligned to Egypt’s event-driven model, ensuring the data you send outward is complete, consistent, and inspection-ready.
See the Product Tour:
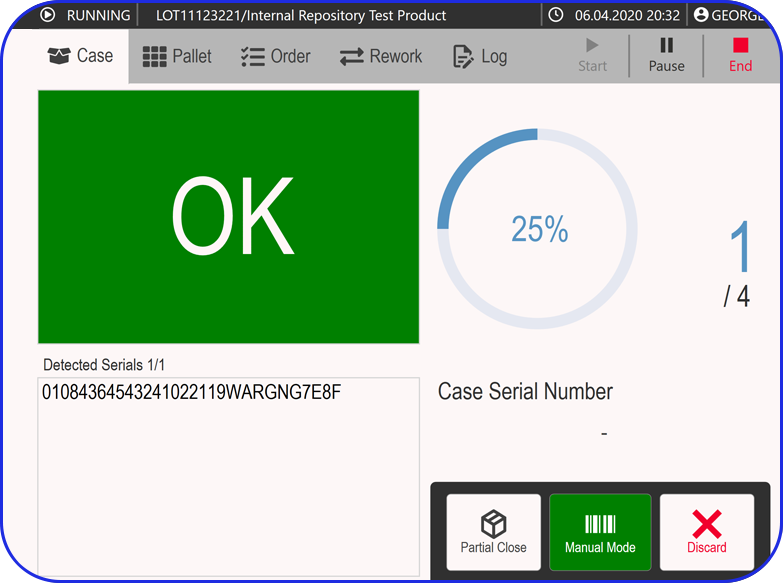
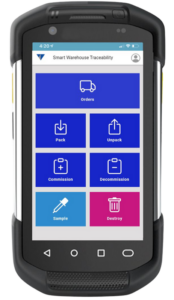
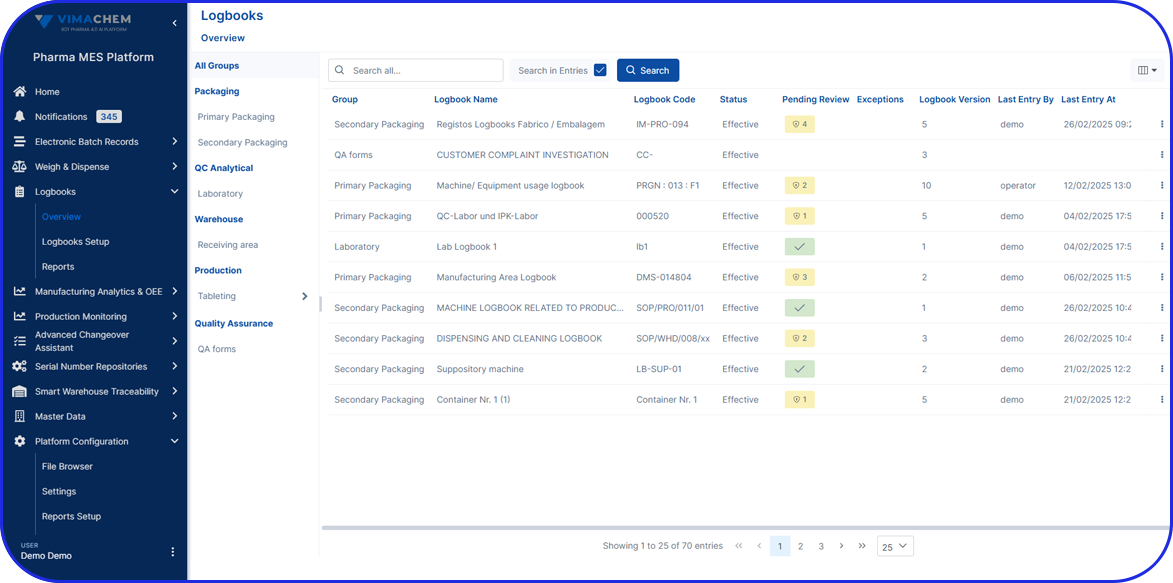
Vimachem Smart Warehouse Traceability (SWT)
Egypt’s compliance doesn’t stop at the packaging line, especially once aggregation is involved. Vimachem’s Smart Warehouse Traceability (SWT) ensures warehouses can handle aggregated stock with confidence. SWT supports Egypt’s readiness by enabling scan-driven receiving and shipping that protects aggregation integrity by maintaining the SSCC-to-child-pack relationship throughout logistics operations. It also enables serialization workflows for distributors and 3PLs, ensuring physical stock movements remain aligned with the traceability record.
Looking Ahead to 2026
Egypt’s Track & Trace enforcement timeline is close: February 2026 for finished imports and August 2026 for local production/packing. Companies that act early can avoid rushed implementation and build a traceability capability that supports both compliance and stronger supply chain visibility. With Vimachem’s serialization solutions, pharmaceutical companies can meet Egypt’s requirements efficiently and turn a regulatory obligation into a more transparent, resilient supply chain.