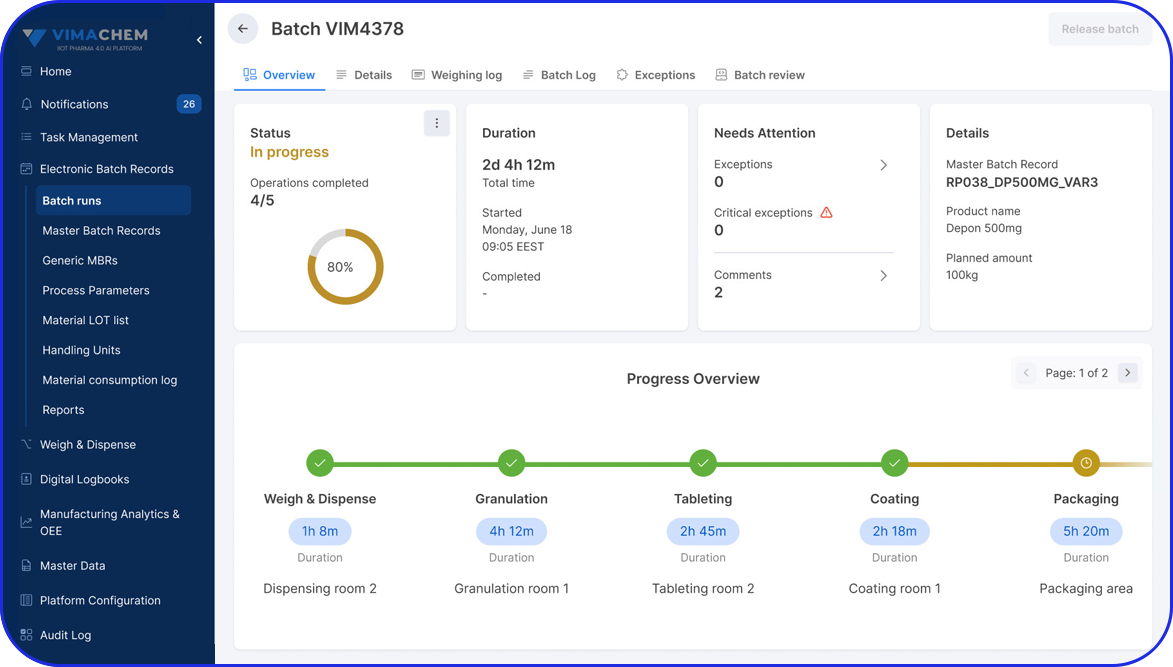
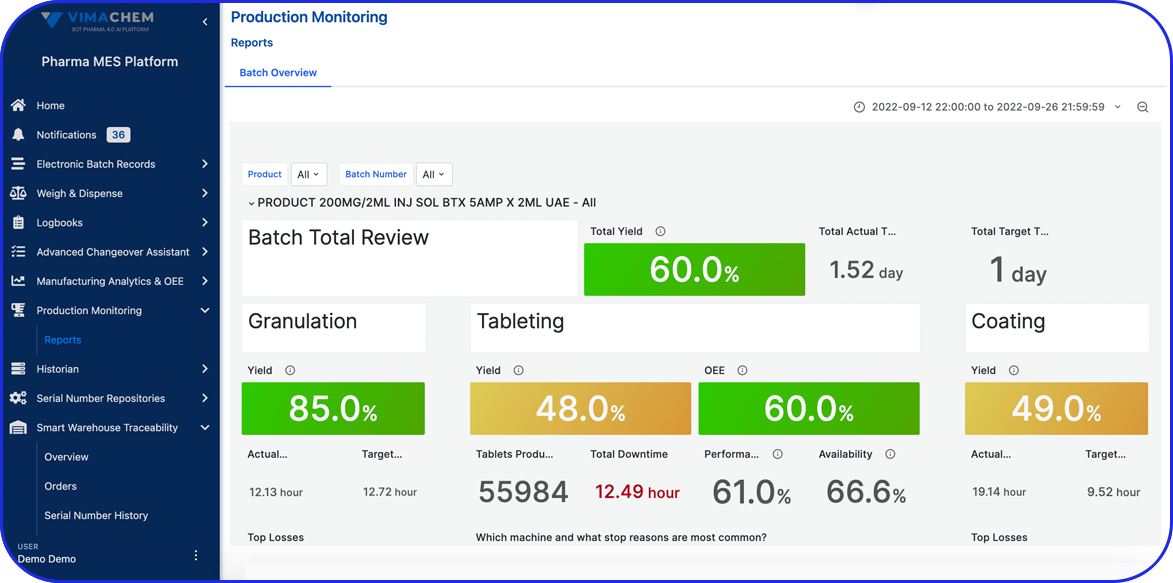
Accelerating Value Through Digitization: A CDMO Case Study on Parallel ERP & eBR Implementation
Accelerating Value Through Digitization: A CDMO Case Study on Parallel ERP & eBR Implementation

Emma Hanley
Pharma Digitization Manager
ERP & EBR Go live readiness:
Up to 50% faster
Revenue Realization:
Up to 18 months earlier
Modern pharma manufacturers can no longer afford the traditional waterfall approach to digital transformation.
For more than a decade, many pharma and biotech organizations have treated ERP upgrades and electronic batch record (eBR) programs as strictly sequential initiatives. The prevailing assumption was that ERP needed to be fully implemented, stabilized, and handed over before any eBR work could begin.
That mindset is now outdated.
Leading manufacturers are increasingly choosing to run ERP and eBR initiatives in parallel. When executed correctly, this approach accelerates time to value, reduces project risk, improves systems integration, reduces validation effort, and avoids years of unnecessary delay.
At Vimachem, we see this shift clearly across the market and have guided multiple clients through successful parallel programs. Recent projects demonstrate that functional scoping, process design, and technical integration can progress simultaneously without compromising GMP compliance, data integrity, or readiness.
Case Study: Parallel ERP and eBR Execution in a Biologics Contract Development and Manufacturing Organization (CDMO)
In one recent biologics CDMO project, operations leadership made a deliberate strategic decision from the outset to run ERP integration and eBR functional design in parallel. The goal was not simply to move faster on paper, but to ensure that technical foundations were established early so functional work could progress without later disruption.
From the first weeks of the program, the teams:
- Established standard, out‑of‑the‑box APIs between the ERP system and the eBR platform using a predefined integration library rather than custom development.
- Defined which production data would originate in ERP and how it would be consumed during execution in eBR.
- Validated real execution scenarios early, rather than waiting until late-stage integration testing.
Using standard integration components, the teams exchanged data such as:
- Material masters and material versions.
- Batch numbers and production orders.
- Critical Process Parameters (CPPs), which are typically managed in the eBR/MES layer (in Vimachem), but in this project were deliberately maintained as master data in the ERP and reused by eBR during execution.
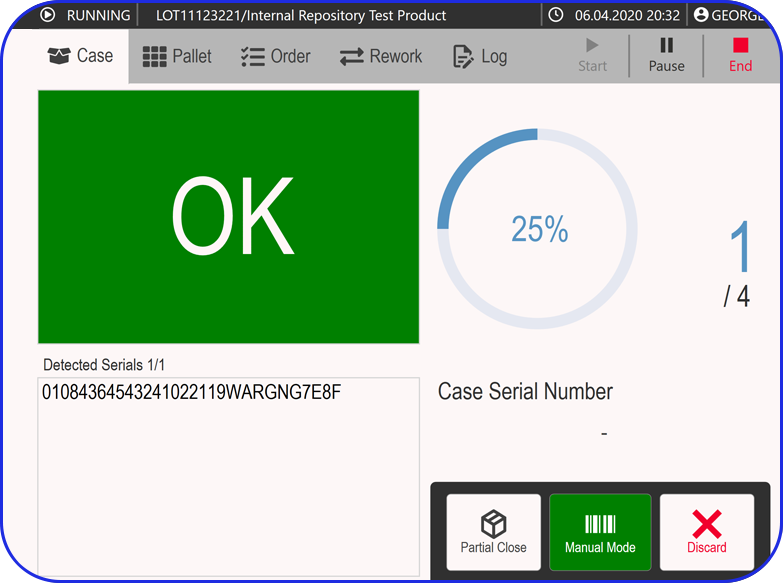
- Status updates and execution confirmations from eBR.
Importantly, ERP was confirmed as the single source of truth for master data. Master data was created and governed in ERP first, and immediately reused within eBR process designs.
This allowed the teams to validate, within the first two to four weeks:
- Data ownership across systems.
- Data formats and transformation rules.
- Error handling and exception management.
- API behavior under real execution conditions.
As a result, both the technical integration stream and the functional eBR design stream matured together. This eliminated the rework typically seen in sequential projects and significantly accelerated readiness for the first stable functional design.
Why Parallel Execution Is Becoming the New Standard

1. ERP and eBR are Interconnected, Not Sequentially Dependent
A common misconception is that eBR implementation must wait until ERP data structures and master data are finalized.
In reality, the systems serve different but complementary purposes:
- ERP defines material masters, material versions, batch numbers, and CPP master data.
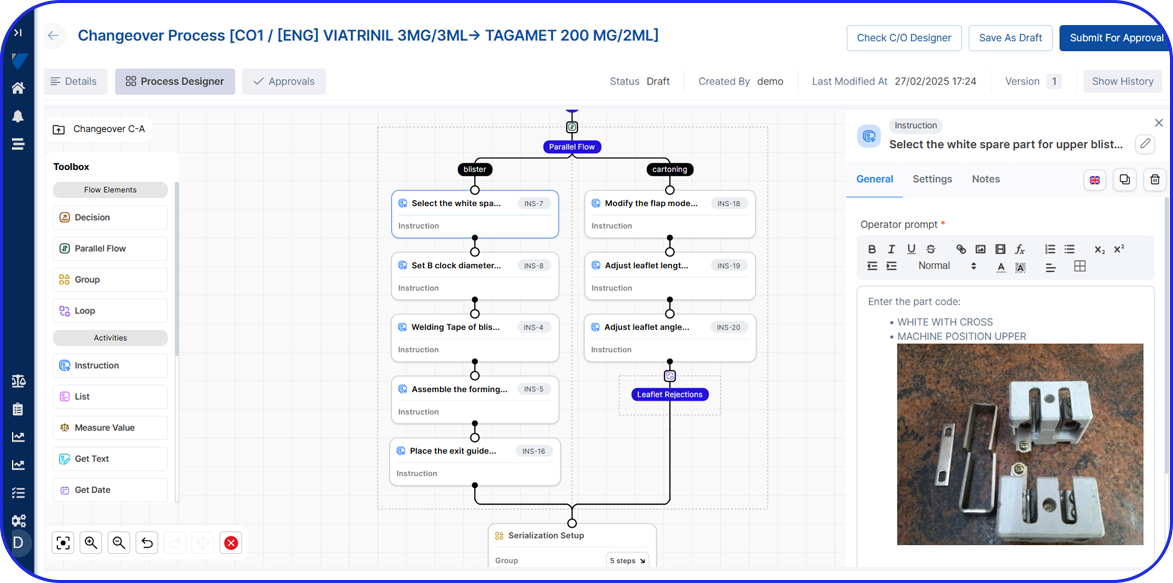
- eBR defines how manufacturing is executed, including step-by-step process logic, electronic master batch records (eMBRs or GMBRs), and operator instructions on the shopfloor.
While these systems are connected through integration, they are not structurally dependent on one another.
A parallel approach allows:
- MS&T and production teams to begin eBR process design and GMBR development immediately.
- ERP teams to refine master data models and parameters on their own timeline.
- Integration teams to progress as soon as initial API expectations are defined.
The result is no idle time and significantly faster time to value.
2. Parallel Execution Optimizes Resource Availability Rather Than Duplicating It
One of the most common objections to parallel execution is resource availability. Organizations often assume that running ERP and eBR at the same time will duplicate effort and overload teams.
In practice, the opposite is true.
Different roles are involved at different stages:
- ERP programs primarily rely on IT, finance, supply chain, and master data teams.
- eBR programs rely heavily on Manufacturing Science & Technology (MS&T), Production, and Quality.
These teams do not need to wait for one another. MS&T can design processes, define CPPs, and structure batch records independently of ERP rollout timing.
A real-world example highlights the risk of not running in parallel. At a mid-sized European pharmaceutical manufacturers, the eBR initiative was paused while the organization focused on upgrading SAP and cleaning master data. The pause was not caused by technical constraints, but by limited organizational bandwidth. As a result, the value of eBR was delayed significantly, despite the work being largely independent.
Parallel execution mitigates this risk by:
- Allowing specialized teams to work concurrently, not redundantly.
- Avoiding long idle periods for MS&T and Quality.
- Preventing digital execution initiatives from being blocked by ERP timelines.
3. Early Technical Integration De-Risks the Entire Program
In the CDMO project, leadership insisted that ERP-eBR integration start immediately, in parallel with functional scoping.
Using a standard integration library, the team validated early:
- Which data needed to be exchanged.
- How data would be exchanged (APIs rather than file-based transfers).
- What format the data should be registered in.
- Which system owned each data element across its lifecycle.
For example:
- Materials, material versions, and CPPs were mastered in ERP.
- eBR consumed this data during execution without redefining it.
- Execution results and deviations flowed back for traceability.
This early validation avoided a common late-stage risk: revisiting hundreds of ERP material records to update a single missing or incorrectly modeled attribute, an issue another customer previously experienced during sequential execution.
By addressing integration questions early:
- Assumptions were tested sooner.
- Technical uncertainty was removed from later validation phases.
- Integration supported validation rather than delaying it.
4. Parallel Execution Supports Agile, Sprint-Based Implementation
Running ERP and eBR in parallel enables a truly agile delivery model.
Rather than waiting for ERP completion, teams can:
- Deliver eBR functionality in sprints.
- Progress ERP configuration and integration incrementally.
- Continuously validate assumptions as systems mature together.
- Reduce implementation timelines by testing, developing, and deploying across both systems concurrently.
This drastically reduces overall implementation time and accelerates time to value.
In a market where digital capabilities are advancing rapidly, pharma companies cannot afford to wait two years for ERP stabilization before digitizing shopfloor execution.
5. Parallel Programs Improve Change Management and Data Ownership Clarity
When systems are implemented sequentially, uncertainty often arises around where data should live and who owns it.
Parallel execution resolves this earlier:
- ERP users adapt to new master data structures.
- eBR users adapt to digital workflows and instruction management.
- Data ownership decisions are clarified upfront.
When users know early which system owns which data, adoption improves and resistance decreases.
6. Parallel Execution Reduces Validation Effort
Contrary to common fears, parallel programs often simplify validation.
Because:
- Interfaces are defined earlier.
- Assumptions are tested incrementally.
- Documentation matures continuously.
- Fewer late changes occur during OQ and PQ.
Validation risk increases when too many elements converge at the end of a program. Parallel execution spreads complexity over time and reduces pressure during validation phases.
7. Parallelization Builds the Shopfloor Digital Foundation Earlier
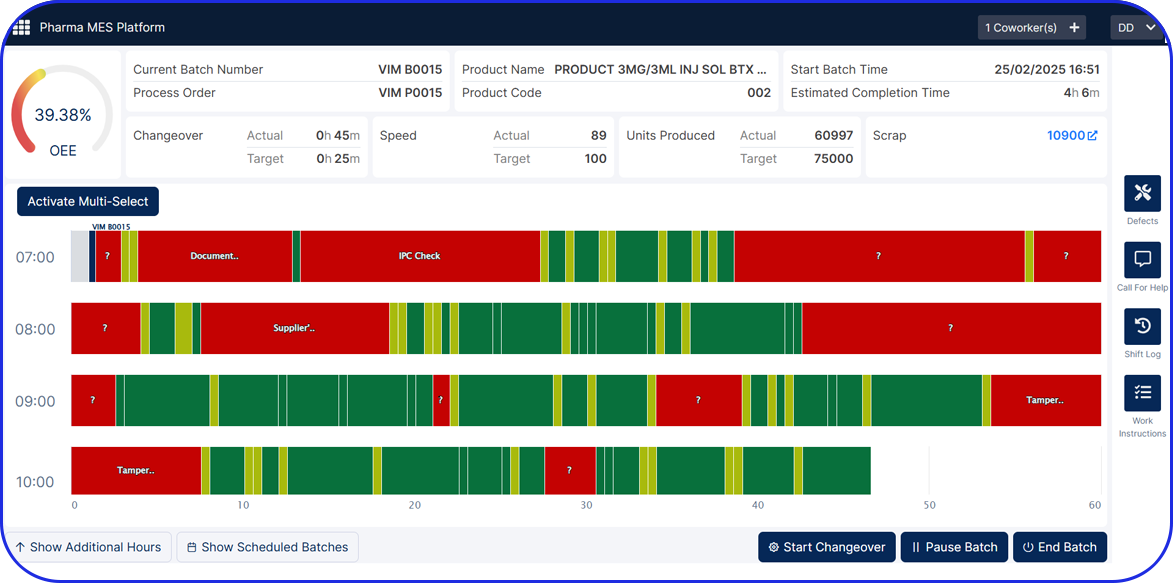
ERP systems alone rarely capture shopfloor reality.
They do not manage:
- Actual execution parameters.
- Equipment states and alarms.
- Step-level deviations.
- What really happened during production.
eBR, together with eLogbooks, OEE, and IIoT, forms the operational truth layer of manufacturing. Building this layer in parallel prevents a common trap: ERP goes live, but production continues to run on paper and spreadsheets because execution was never digitized.
This undermines both ERP adoption and ROI.
A Practical ERP Transition Scenario Enabled by Parallel Execution
Parallel execution also supports ERP transitions.
A company may start eBR integrated with ERP system #1 using standard APIs and a clearly defined data model. Later, during a side-by-side rollout of ERP system #2, the same integration logic can be reused.
The result:
- Minimal disruption to eBR.
- No rework of execution logic.
- A controlled and validated switch at the ERP layer.
Want to Learn How to Effectively implement eBR in line with your ERP?
Conclusion: Parallel Beats Sequential
Modern MES and digital manufacturing architectures are designed for concurrency.
Organizations that embrace parallel ERP and eBR execution:
- Shorten implementation timelines.
- Improve integration quality.
- Reduce validation and project risk.
- Clarify data ownership earlier.
- Increase user adoption.
- Achieve earlier digital ROI.
Parallel execution is no longer an ambitious approach. It is becoming the new standard for pharma manufacturers who want to accelerate digital transformation without compromising compliance.
Running ERP and eBR in parallel positions organizations to lead digital manufacturing initiatives rather than waiting years for sequential programs to finish.