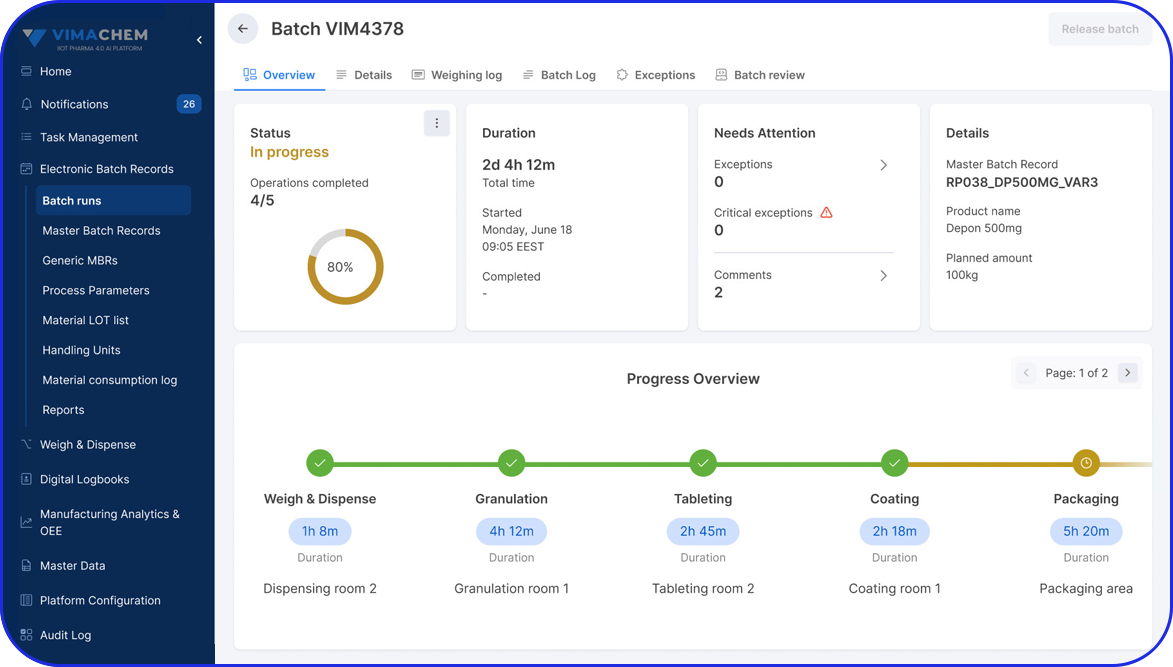
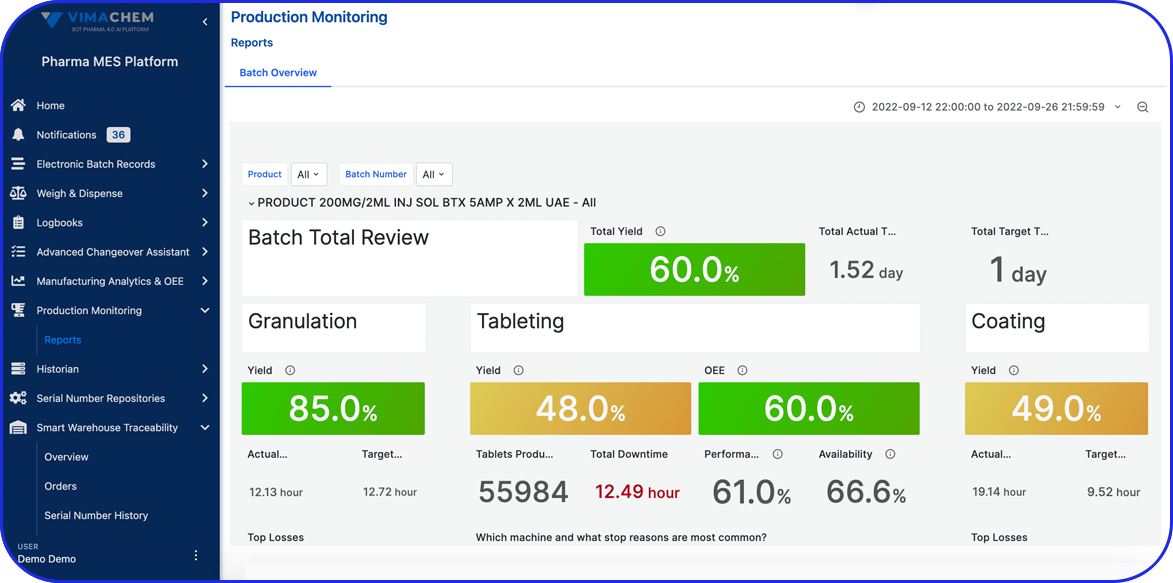
Product Tour
Benefits of Vimachem’s OEE Software
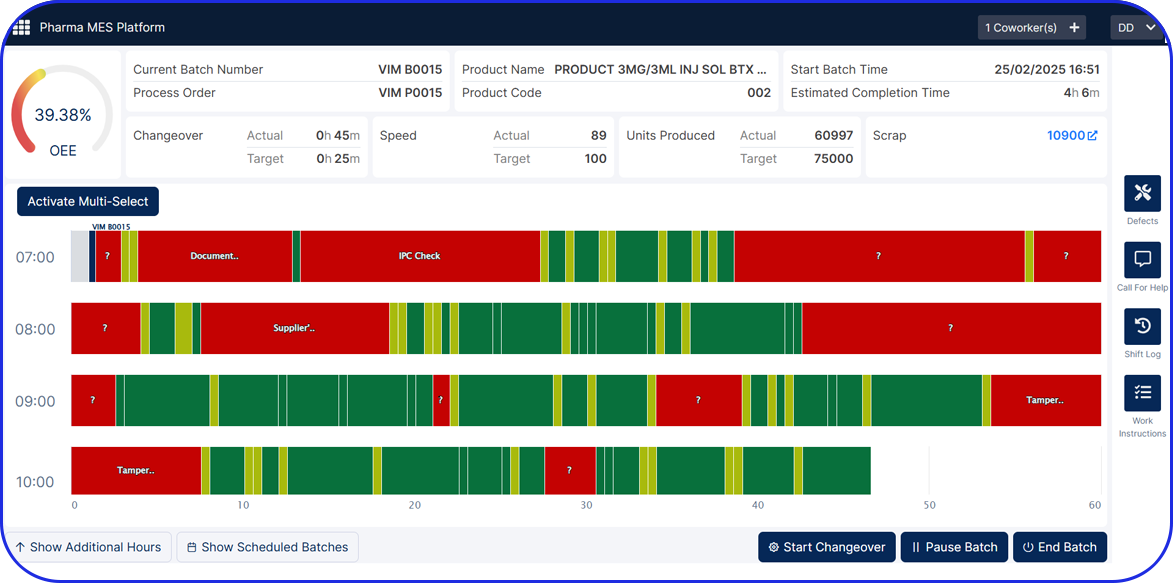
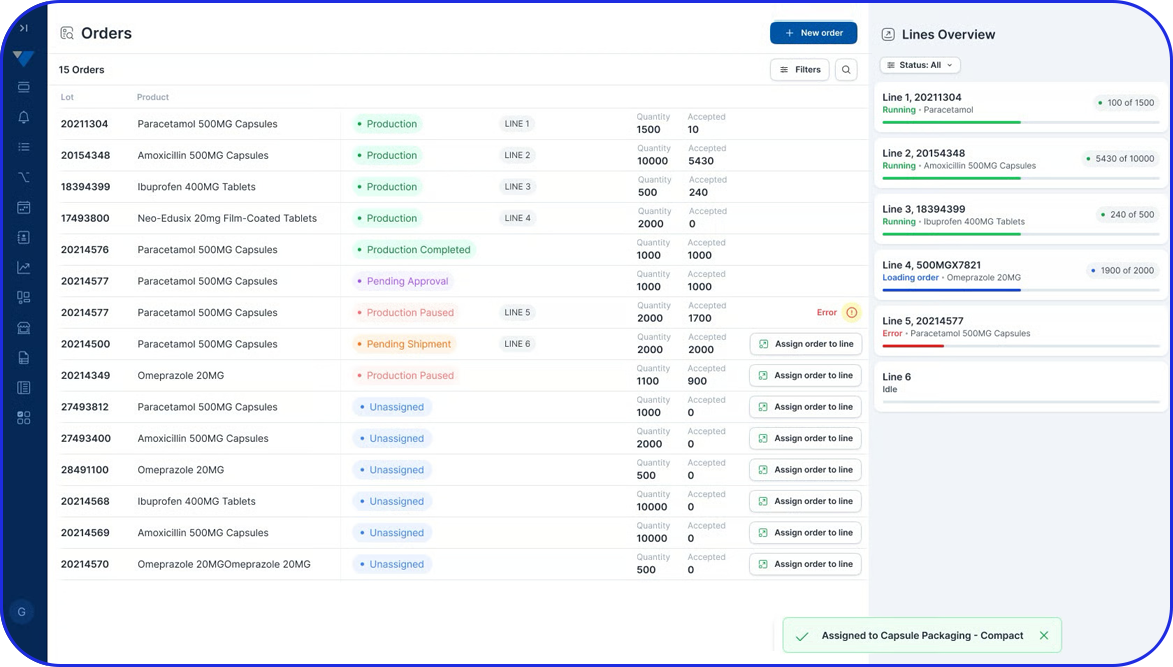
Optimize Performance with Real-Time OEE Monitoring
Disconnected systems and limited visibility make it difficult for pharma manufacturers to respond quickly to production inefficiencies. Vimachem’s Manufacturing Analytics & OEE module provides real-time insights into critical KPIs - enhancing performance monitoring, equipment utilization, and compliance with GMP and operational excellence standards.
Pharma-Specific OEE Reporting Software
Implementation delays due to lengthy validation cycles can stall digital transformation and increase costs.
Vimachem’s pre-validated solution includes a comprehensive, out-of-the-box validation package -reducing deployment timelines by at least two months while ensuring full compliance from the initial rollout.
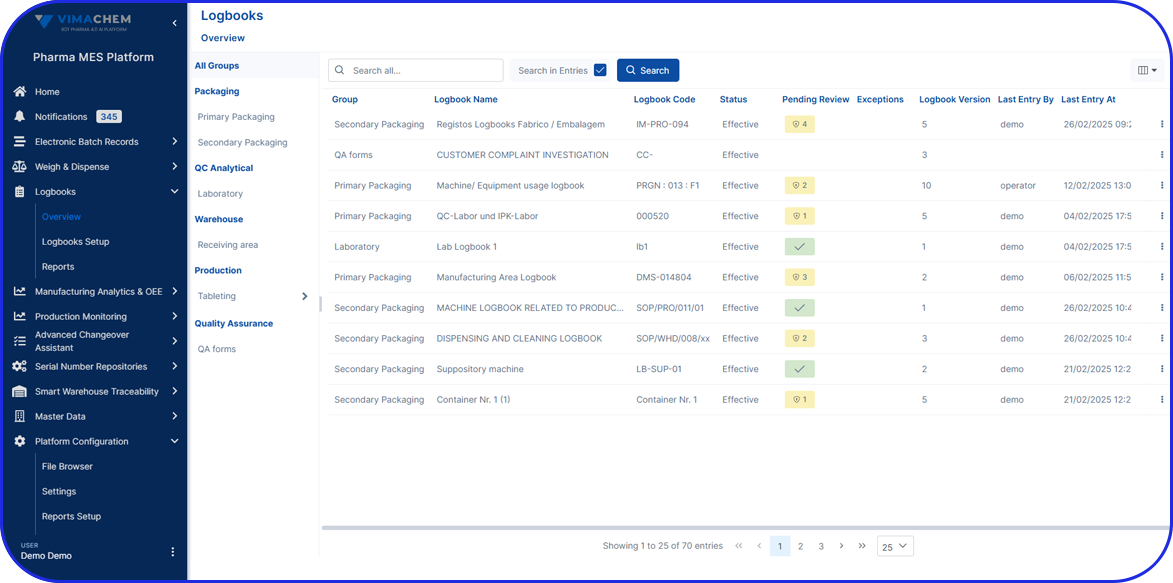
Seamless OEE System Integration & Data Collection
Manual data entry and fragmented systems can lead to operational blind spots and increased risk. Vimachem’s MA&OEE module integrates effortlessly with your ERP, shop floor industrial equipment, and RFID systems—providing a unified, real-time view of production performance without the need for additional infrastructure or complex IT setup.
See Vimachem’s MA & OEE in Action
OEE Software for Pharma
Built for Pharma and Biotech Teams to Drive Performance, Compliance, and Operational Excellence
Pharmaceutical and biotech manufacturers need to maximize throughput while maintaining quality and meeting regulatory requirements. Vimachem’s Manufacturing Analytics & OEE module provides the insights and tools needed to improve efficiency, standardize operations, and ensure compliance across all production sites.
Vimachem’s Manufacturing Analytics & OEE solution empowers pharma and biotech teams to drive continuous improvement, ensure compliance, and unlock higher productivity, delivering smarter, faster, and more efficient manufacturing across all sites.
Key Features of Our OEE Software
- A GAMP5 Category 4, GMP-compliant platform purpose-built for pharmaceutical and biotech environments.
- Real-time monitoring of performance, downtime, and changeovers to identify and address inefficiencies.
- A user-friendly, intuitive interface with pharma-specific dashboards designed for operators, supervisors, and managers with no coding required.
- Automated alerts that highlight production issues and enable fast, informed responses.
- Seamless integration with ERP systems, machines, and RFID readers to unify data across operations.
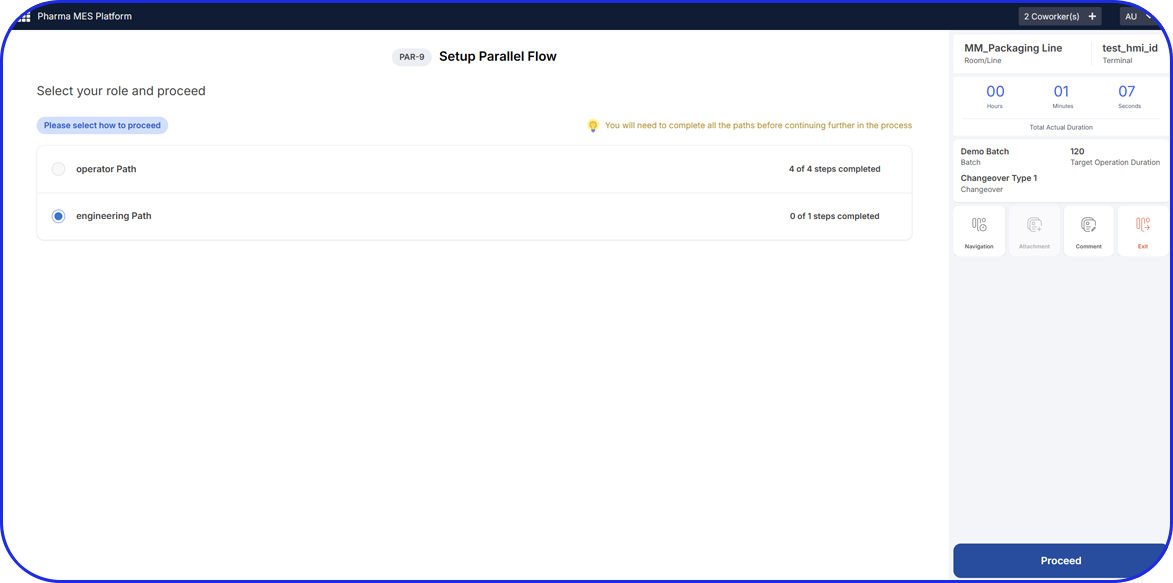
Machine Connectivity for Real-Time OEE Data Collection
With deep expertise in machine and data protocols, Vimachem’s automation and software engineers have completed machine data connectivity projects in over 20 countries worldwide.
Our proven capabilities in machine data acquisition, combined with our deep technical knowledge of the pharmaceutical environment makes us the ideal partner for connecting machines across the pharmaceutical shop floor.

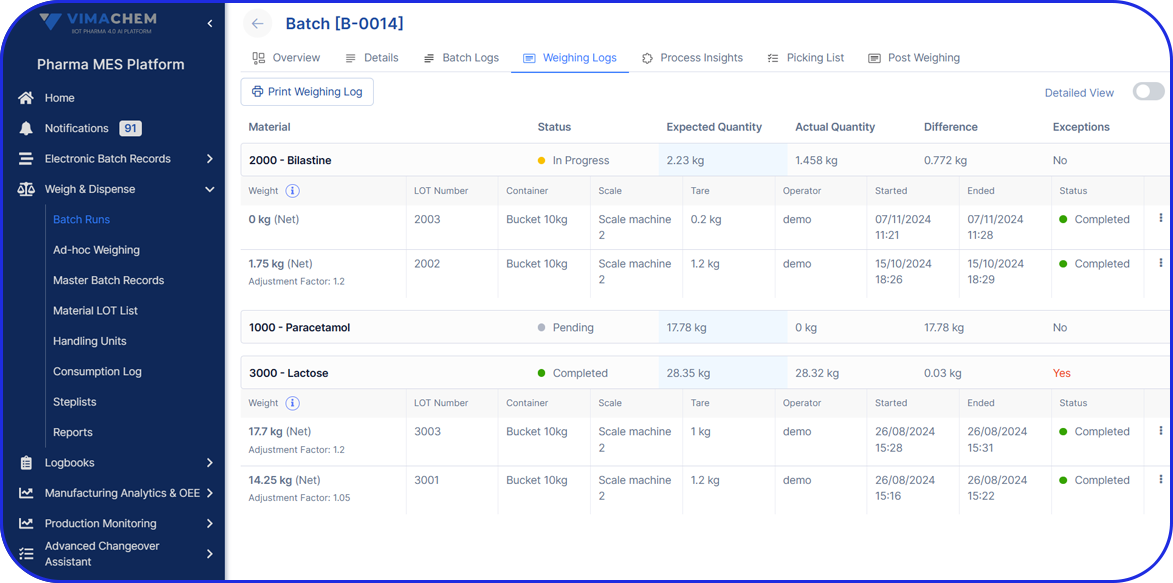
Real-Time OEE Software for Pharma
Plug-and-Play Real-Time OEE Monitoring, Live in Days
Begin fully automating OEE calculations in just hours - no invasive setup, no downtime. Real insights and fast results.
Empower Shop-Floor Teams with OEE Tracking Tools
AI-powered, operator-centric tools streamline shift handovers, instructions, and reporting - driving daily performance gains.
Data-Driven Decisions with OEE Reporting & Analytics
Track KPIs and improvement plans with PDCA-based dashboards that simplify operational excellence initiatives.
Real-Time OEE Alerts & Downtime Monitoring
Minimize unplanned downtime with automated alerts that highlight root causes and guide rapid resolution.
GMP-Ready OEE Software for Pharma & Biotech
A GAMP5 Category 4, GMP-compliant solution to compare and evaluate manufacturing data across multiple facilities through a single, unified interface.
Digital Continuous Improvement Driven by OEE
Identify, assign and monitor continuous improvement routines and tasks and improve efficiency while reducing costs and employee turnover.
Our journey with Vimachem has been truly rewarding. From connecting machines to installing the Vimachem MES platform, we rapidly integrated 6 packaging lines without any production disruptions.George Vlassopoulos, Head of Engineering, Elpen
GxP & Compliance Features for Pharma OEE Software
User-Level Permissions & Security
Role-based access, AD integration, and reduced IT admin effort.
Verified Data Integrity
System validation, input checks, and secure access control.
Electronic Records & Signatures
Fully compliant with FDA 21 CFR Part 11 and EU GMP Annex 11.
Comprehensive Digital Records
Full traceability of shop floor actions and equipment usage.
Audit Logs
Track every system activity for compliance and troubleshooting.
Flexible Data Exports
Export data in XLSX/CSV formats manually or automatically.
Scalable OEE Solutions with Modular Add-Ons
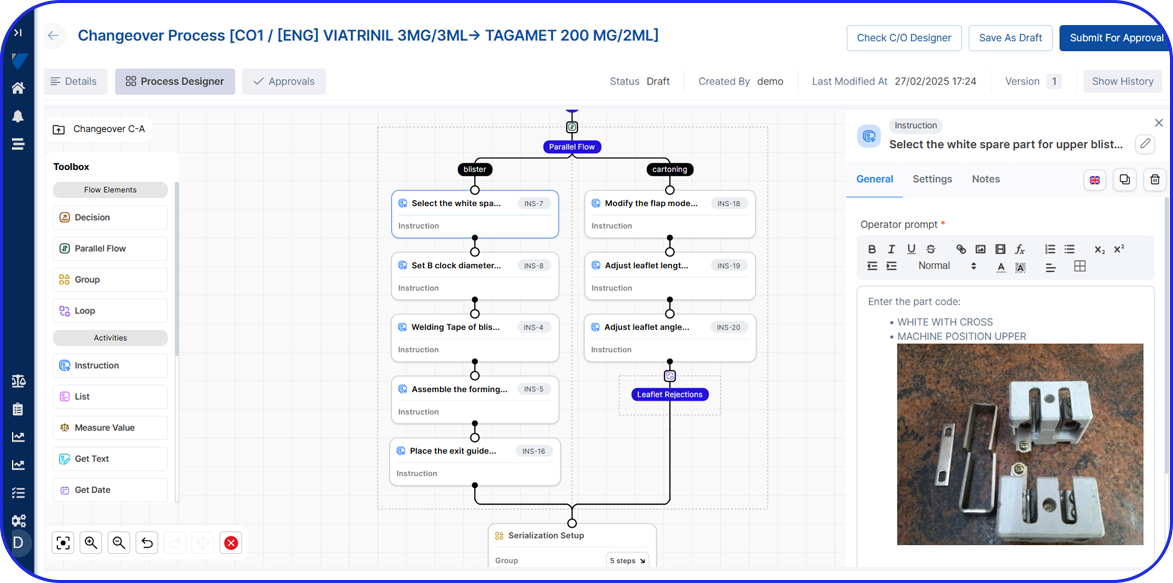
Extend Vimachem’s OEE platform with modular add-ons such as changeover instruction & analytics, electronic batch records, and a serialization site manager to close visibility gaps across packaging and production.
Changeover Instruction & Analytics
Electronic Batch Record
Combine with eBR to streamline packaging records and ensure full compliance.
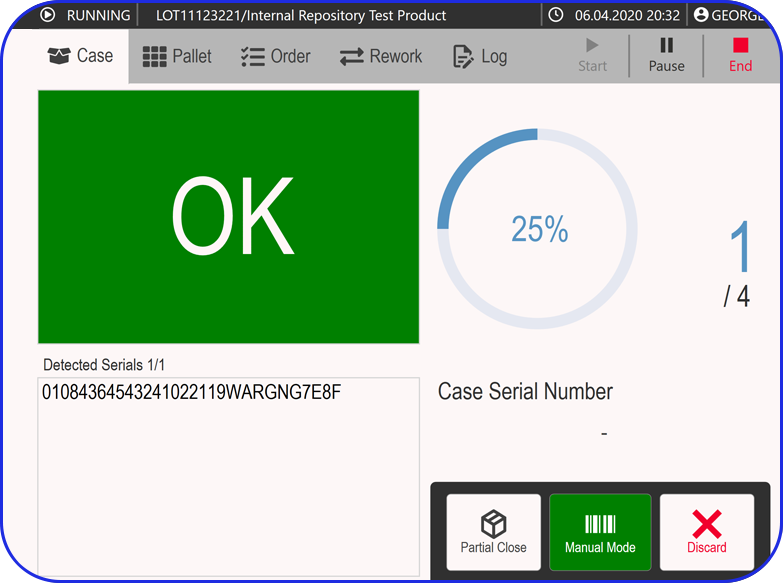
Serialization Site Manager
Track serialization efficiency and monitor packaging line performance with a single machine connection.
Contact us
FAQs on OEE Software & Pharma Manufacturing Analytics
What is OEE software and what does it do?
OEE software (overall equipment effectiveness software) measures Availability, Performance and Quality to quantify how effectively equipment is producing. It turns raw shop-floor signals into real-time OEE monitoring, automated OEE calculation, dashboards and OEE reporting so teams can prioritize fixes and improve throughput.
How does OEE software collect machine data?
Modern OEE data collection software connects directly to PLCs, HMIs, historians and edge devices via OPC UA, MQTT, native PLC drivers or APIs. It also accepts RFID/scanners and manual reason-code inputs where needed, normalizes tags, and timestamps events for accurate calculation and auditability.
How is OEE calculated in the system?
OEE = Availability × Performance × Quality. The platform automatically derives Availability (run time vs scheduled time), Performance (actual vs ideal cycle time) and Quality (good units ÷ total units) from captured events and machine signals, producing drill-downs by line, product, shift or SKU.
Can OEE software detect and reduce downtime?
Yes. OEE tracking and downtime monitoring capture stops, micro-stops, and slow cycles, attach standardized reason codes, trigger real-time alerts, and provide Pareto/RCA tools so teams can eliminate top causes of unplanned downtime and recover lost throughput faster.
Will OEE software integrate with our ERP/MES/SCADA?
Absolutely. OEE solutions typically include connectors and APIs to integrate ERP, MES, SCADA, and historian systems so production orders, batch/lot data and master data enrich OEE metrics giving context for validated reporting and CI initiatives.
Is OEE software suitable for multi-site operations?
Yes. Digital OEE solutions scale across lines and facilities by normalizing tags, standardizing reason codes, and centralizing dashboards, enabling site-to-site comparisons, roll-up reporting, and multi-site benchmarking.
How long does it take to see value and ROI from OEE?
You start getting visibility immediately after machine connectivity and initial configuration; measurable ROI depends on the size of losses identified. Most customers realize first improvements (reduced micro-stops, faster changeovers) within weeks once the top causes are addressed, and broader ROI as CI routines are closed.
What reporting and export options are available?
OEE reporting software typically offers scheduled scorecards, drill-down dashboards, PDF/Excel (XLSX/CSV) exports, and BI integrations so leaders and CI teams can track trends, audits, and KPI-based actions.
How is Pharma Manufacturing Analytics different from standard OEE?
Pharma analytics combines real-time OEE monitoring with GxP controls: batch and lot linking, electronic records, validated data pipelines, audit trails and role-based security. It’s designed for GMP environments where traceability, data integrity and regulatory evidence are required in addition to production optimization.
How does Vimachem’s solution support compliance and validation?
Vimachem’s MA & OEE includes GAMP5-aligned features (validated data flows, system validation packages), FDA 21 CFR Part 11 / EU Annex 11-compatible electronic records and eSignatures, audit logs, and secure access controls—helping reduce validation timelines and delivering audit-ready OEE reporting for pharma and biopharma manufacturing.
Ready to Augment your Shop Floor Operations?
Ready to Augment your Shop Floor Operations?
Get started with real-time manufacturing analytics today.